Chemical Bonding & Molecular Structure
Cheatsheet Content
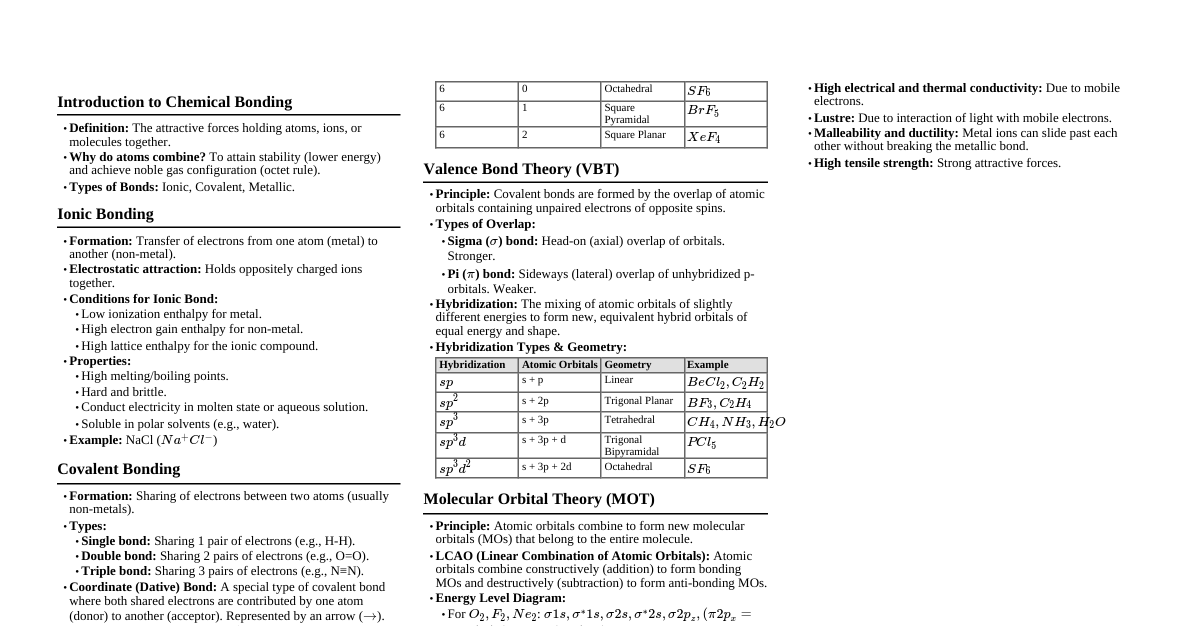
Chemical Bond Introduction Most elements exist as molecules (cluster of atoms). Molecules form if more stable and have lower energy than individual atoms. Chemical Bond: Force holding two or more atoms together in a stable molecule. Involves redistribution of electrons. Process accompanied by energy decrease. Decrease in energy $\propto$ Strength of bond. Molecules are more stable than atoms. Cause of Chemical Combination: Tendency to acquire minimum energy When atoms approach, nucleus of one attracts electrons of another. Repulsion exists between two nuclei and between electrons of both atoms. If net attraction occurs, total energy of the system decreases, forming a chemical bond. Attraction $\propto$ 1/Energy $\propto$ Stability. Bond formation is an exothermic process ($\Delta H = \text{negative}$). Potential energy (KJ/mol) Distance of separation Bond Energy (Internuclear distance) Classification of Bonds Chemical Bonds Strong Bond (Interatomic) (Energy $\ge$ 200 KJ/mole) Weak Bond (Intermolecular) (Energy $ Types Ionic, Covalent, Co-ordinate, Metallic Hydrogen, Van der Waals Ionic or Electrovalent Bond Formed by complete transfer of one or more electrons from one atom to another. Electropositive (metal) atom loses electrons. Electronegative (non-metal) atom gains electrons. Electrostatic force of attraction between cation and anion forms the bond. Electronegativity difference $\propto$ Nature of ionic bond. (e.g., Group IA & VIIA elements form max ionic compounds). Total number of electrons lost or gained is called electrovalency. Example: Formation of NaCl Na (2,8,1) + Cl (2,8,7) $\rightarrow$ Na$^+$ (2,8) + Cl$^-$ (2,8,8) Electrovalency of Na = 1, Cl = 1. Example: Formation of MgCl$_2$ Mg (2,8,2) + 2Cl (2,8,7) $\rightarrow$ Mg$^{2+}$ (2,8) + 2Cl$^-$ (2,8,8) Electrovalency of Mg = 2, Cl = 1. Representation of Formula of Compounds Write symbols of ions side by side (positive ion left, negative ion right) as A$^x$B$^y$. Write electrovalency as superscript. Apply Criss-cross rule to get formula A$_y$B$_x$. Example: Calcium Chloride: Ca$^{2+}$Cl$^1$ $\rightarrow$ CaCl$_2$. Lattice Energy (L.E.) Energy released when one mole of crystal lattice forms from gaseous ions, or energy required to break one mole of crystal lattice into gaseous ions. Higher lattice energy $\rightarrow$ Greater stability/strength of ionic compound. Factors Affecting Lattice Energy Magnitude of Charge: $U \propto q_1 q_2$ (charge on ions). Lattice energy $\propto$ Magnitude of charge. NaCl $ Size of Cation: Lattice energy $\propto \frac{1}{r}$. LiCl $>$ NaCl $>$ KCl $>$ RbCl $>$ CsCl (for constant anion, lattice energy decreases as cation size increases). Conditions for Forming Ionic Bonds Ionisation Energy (I.E.): Amount of energy required to remove an electron from outermost orbit of an isolated gaseous atom to form a cation. (Energy absorbed). Lesser I.E. $\rightarrow$ Greater tendency to form cation. Example: Na$^+$ $>$ Mg$^{2+}$ $>$ Al$^{3+}$ (Cation formation tendency). Electron Affinity (E.A.): Amount of energy released when an electron is added to an isolated gaseous atom to form an anion. (Energy released). Higher E.A. $\rightarrow$ Greater tendency to form anion. Example: Cl $>$ F $>$ Br $>$ I. Lattice Energy (U): Higher lattice energy $\propto$ strength of ionic bond. Covalent Bond Formed by mutual sharing of electrons between two atoms to complete their octet (except H, which completes its duplet). Shared electron pair has opposite spins and is localized between the two atoms. Sharing can occur in three ways: 2 electrons shared $\rightarrow$ 1 electron pair $\rightarrow$ Single bond ($-$): e.g., H$-$H 4 electrons shared $\rightarrow$ 2 electron pairs $\rightarrow$ Double bond ($=$): e.g., O$=$O 6 electrons shared $\rightarrow$ 3 electron pairs $\rightarrow$ Triple bond ($\equiv$): e.g., N$\equiv$N Orbital Concept of Covalent Bond One orbital accommodates max 2 electrons with opposite spins ($\uparrow\downarrow$). Half-filled or unpaired electron orbital accepts one electron from another atom to complete its orbital. Tendency to complete orbital or pair electrons is essential for covalent bond. Completion of octet is NOT always essential. Covalency: Number of covalent bonds an atom forms in a molecule (ground or excited state). Variable Valency in Covalent Bonds Shown by elements with empty orbitals in outermost shell. Lone pair electrons can be excited to form maximum unpaired electrons, showing maximum covalency in excited state. Elements from 3rd, 4th, 5th, 6th periods can show variable valencies due to vacant d-orbitals (with highly electronegative elements like F, O, Cl). Energy for electron excitation is called promotion energy/excitation energy. Promotion Rule: Excitation of electrons occurs in the same orbit. Example: Nitrogen (Ground State) $2s^2 2p^3: \uparrow\downarrow \quad \uparrow\uparrow\uparrow$ Covalency = 3 (e.g., NCl$_3$). Nitrogen has no excited state, so NCl$_5$ does not exist (due to absence of vacant d-orbital). Example: Phosphorus (Ground State) $3s^2 3p^3: \uparrow\downarrow \quad \uparrow\uparrow\uparrow$ Covalency = 3 (e.g., PCl$_3$). Phosphorus (Excited State) $3s^1 3p^3 3d^1: \uparrow \quad \uparrow\uparrow\uparrow \quad \uparrow$ Covalency = 5 (e.g., PCl$_5$). PCl$_3$ and PCl$_5$ both exist due to presence of 3d orbitals. Note NCl$_3$ exists, NCl$_5$ doesn't exist (no d-orbitals in N). OF$_2$ exists, OF$_4$ and OF$_6$ don't exist (no d-orbitals in O). SF$_4$ and SF$_6$ exist (d-orbitals present in S). Example: Sulphur (Ground State) $3s^2 3p^4: \uparrow\downarrow \quad \uparrow\downarrow\uparrow\uparrow$ Covalency = 2 (e.g., SF$_2$). Sulphur (1st Excited State) $3s^1 3p^3 3d^2: \uparrow \quad \uparrow\uparrow\uparrow \quad \uparrow\uparrow$ Covalency = 4 (e.g., SF$_4$). Sulphur (2nd Excited State) $3s^1 3p^3 3d^2: \uparrow \quad \uparrow\uparrow\uparrow \quad \uparrow\uparrow$ (Incorrect as $3s^1 3p^2 3d^3$ needed) Covalency = 6 (e.g., SF$_6$). Variable covalency of S is 2, 4, & 6. Co-ordinate Bond (Dative Bond) Covalent bond where the shared electron pair comes from one atom. Donor atom (Lewis base) provides the electron pair. Acceptor atom (Lewis acid) accepts the electron pair. Also called donor-acceptor bond. Example: NH$_3$ + BF$_3$ NH$_3$ (Lewis base) donates a lone pair to BF$_3$ (Lewis acid, electron deficient). H$_3$N: $\rightarrow$ BF$_3$ Necessary Conditions Donor atom must have a complete octet and at least one lone pair. Acceptor atom must have a vacant orbital to accommodate the lone pair. Protonation (Addition of H$^+$ ion) Example: NH$_4^+$ (Ammonium ion): NH$_3$ + H$^+$ $\rightarrow$ NH$_4^+$ Example: H$_3$O$^+$ (Hydronium ion): H$_2$O + H$^+$ $\rightarrow$ H$_3$O$^+$ Other Examples of Co-ordinate Bond O$_3$: O=O$\leftarrow$O $\leftrightarrow$ O$\rightarrow$O=O N$_2$O$_5$: O$_2$N$-$O$-$NO$_2$ H$_2$SO$_4$: O=S(OH)$_2$ Compounds with Ionic, covalent and co-ordinate bonds: NH$_4$Cl, Na$_3$PO$_4$, KNO$_3$. Lewis Dot Structures or Lewis Symbols Only outermost shell electrons (valence electrons) participate in chemical combination. Lewis introduced simple notations for valence electrons. Lewis Symbols for Atoms and Monoatomic Ions Number of dots around the symbol represents the number of valence electrons. Examples: $\cdot$Li, $\cdot$Be$\cdot$, $\cdot$B$\cdot$, $: \ddot{F} \cdot$, $: \ddot{Ne}:$ Lewis symbol for anions: $: \ddot{F}:^-$ How to Draw Lewis Electron Dot/Bond Line Structures (Inorganic Covalent Compounds) Identify Central Atom: Usually the least electronegative atom. Hydrogen cannot be central atom (covalency = 1). Sometimes the atom present in lesser number. If electronegativity/number is inconclusive, the atom in the central position of the formula. Examples: CH$_4$, NH$_3$, H$_2$O, CO$_2$, SO$_2$, SO$_3$, PCl$_3$, OCl$_2$, (SCN)$^-$ Surrounding atoms are directly bonded to the central atom. Arrange surrounding atoms around central atom and form single bonds first. Ensure octet for all surrounding atoms. If not, form double or triple bonds. Represent lone pairs on central and surrounding atoms. Ensure octet for all surrounding atoms is complete. If central atom is from 2nd period, it can have $\le$ 8 electrons ($\le$ 4 bonds). If central atom is from 3rd or lower period, it can have $\ge$ 8 electrons (expanded octet). Verify covalency of central atom. Lewis Structure of Ions Distribute negative charge on surrounding atoms such that octet is incomplete before bond formation. If octet is complete, surrounding atoms attach via coordinate bonds (especially halogens with 7 valence electrons). Example: NO$_3^-$ Incorrect structure with N forming 5 covalent bonds (N is 2nd period element, cannot expand octet). Other Examples CO$_3^{2-}$ BF$_4^-$ PO$_4^{3-}$ Formal Charge Lewis dot structures don't always represent actual shapes. For polyatomic ions, net charge is for the whole ion, not a particular atom. Formal charge can be assigned to each atom. $Q_F = N_A - N_{L.P.} - \frac{1}{2} N_{B.P.}$ $N_A$: Total valence electrons in free atom $N_{L.P.}$ : Total non-bonding (lone pair) electrons $N_{B.P.}$ : Total bonding (shared) electrons Formal charges help select the lowest energy structure. Exception of Octet Rule (Failure) Incomplete Octet / Electron Deficient / Hypovalent Molecules: Central atom has less than 8 electrons. Examples: BF$_3$, AlCl$_3$, BCl$_3$, BeCl$_2$, HgCl$_2$, Ga(CH$_3$)$_3$. Expanded Octet / Electron Efficient / Hypervalent Molecules: Central atom has more than 8 electrons. Examples: PCl$_5$ (10e$^-$), SF$_6$ (12e$^-$), IF$_7$ (14e$^-$). Pseudo Inert Gas Configuration: Cations with 18 electrons in outermost orbit. Examples: Ga$^{3+}$, Cu$^+$, Ag$^+$, Zn$^{2+}$, Cd$^{2+}$, Sn$^{4+}$, Pb$^{4+}$. Odd Electron Molecules: Central atom has an unpaired or odd number of electrons (7e$^-$, 11e$^-$, etc.). Examples: NO, NO$_2$, ClO$_2$. Resonance When a molecule cannot be completely represented by a single Lewis structure, but its properties can be described by two or more different structures with similar energy and atomic positions. The true structure is a resonance hybrid of these contributing (canonical) structures. Resonance stabilizes the molecule (resonance hybrid has lower energy than canonical forms). Resonance averages bond characteristics. Canonical forms have no real existence. Resonance Energy Energy difference between most stable resonating structure and resonance hybrid structure. Rules for Writing Resonating Structures Contributing structures with similar energy contribute equally to the resonance hybrid. Higher energy canonical forms contribute less. Contributing structures must have the same atomic positions, differing only in electron positions. (e.g., H$_3$PO$_3$ structures are not resonating structures). Same number of unpaired electrons. Should not differ much in energy. Negative charge on more electronegative element, positive charge on less electronegative element. Like charges should not reside on adjacent atoms. (e.g., N$_2$O structure with positive charges on adjacent N atoms is less feasible). A resonating structure violating the octet rule will be least stable. Example: CH$_2$=CH$-$Cl $\leftrightarrow$ CH$_2^-$-CH=Cl$^+$ $\leftrightarrow$ CH$_2^+$-CH=Cl$^-$ Bond Order Number of bonds between two atoms. Resonance Structure of Some Molecules/Ions Azide ion, N$_3^-$: Three resonating structures. II and III contribute equally, showing almost double bond character in each N-N bond. Carbonate, CO$_3^{2-}$: Three resonating structures, bond order = 1.33. Benzene, C$_6$H$_6$: Bond order = 1.5. Carboxylate ion, RCOO$^-$: Bond order = 1.5. Valence Bond Theory (VBT) Presented by Heitler & London, extended by Pauling & Slater. Main Points: Covalent bond forms via overlapping of half-filled valence shell orbitals. Orbitals approach from direction of maximum overlapping, giving directional character to bond. Strength of covalent bond $\propto$ extent of overlapping. Extent of overlapping depends on: Nature of Orbitals: As $n$ increases, bond strength decreases (e.g., $1s-2p > 2s-2p > 3s-3p$). p, d, f orbitals are directional $\rightarrow$ more overlapping. s-orbitals are non-directional $\rightarrow$ less overlapping. Directional overlapping gives stronger bonds. (e.g., $p-p > s-p > s-s$). If $n$ is same: $2p-2p > 2s-2p > 2s-2s$. Nature of Overlapping: Co-axial overlapping $\rightarrow$ greater extent of overlapping. Collateral overlapping $\rightarrow$ lesser extent of overlapping. Order of strength (Co-axial): $p-p > s-p > s-s$. Types of Overlapping and Nature of Covalent Bonds Sigma ($\sigma$) Bond: Formed by end-to-end (head-on or axial) overlap along the internuclear axis. s-s overlapping: Two half-filled s-orbitals overlap. s-p overlapping: Half-filled s-orbital and half-filled p-orbital overlap. p-p overlapping: Two half-filled p-orbitals overlap axially. Pi ($\pi$) Bond: Formed by sidewise overlapping of atomic orbitals, with axes parallel and perpendicular to the internuclear axis. Results in two saucer-type charged clouds above and below the plane. Examples: $p_\pi-p_\pi$ overlapping, $p_\pi-d_\pi$ overlapping. Delta ($\delta$) Bond: Covalent bonds where four lobes of one d-orbital overlap with four lobes of another d-orbital. Except $d_{z^2}$, all d-orbitals can form $\delta$ bonds. Table: Atomic Orbitals and Type of Covalent Bond (Internuclear Axis) Atomic Orbitals Internuclear Axis Type of Covalent Bond s + p$_x$ x axis $\sigma$ y axis, z axis X s + p$_y$ y axis $\sigma$ x axis, z axis X s + p$_z$ z axis $\sigma$ x axis, y axis X p$_x$ + p$_x$ x axis $\sigma$ y axis, z axis $\pi$ p$_y$ + p$_y$ y axis $\sigma$ x axis, z axis $\pi$ p$_z$ + p$_z$ z axis $\sigma$ x axis, y axis $\pi$ p$_x$ + d$_{xy}$ y axis $\pi$ p$_z$ + d$_{yz}$ y axis $\pi$ p$_z$ + d$_{zx}$ x axis $\pi$ d$_{xy}$ + d$_{xy}$ x axis, y axis $\pi$ z axis $\delta$ d$_{yz}$ + d$_{yz}$ y axis, z axis $\pi$ x axis $\delta$ d$_{xz}$ + d$_{xz}$ x axis, z axis $\pi$ y axis $\delta$ Strength of Sigma and Pi-Bonds Sigma bonds involve greater overlapping, thus are stronger than pi bonds. Pi bonds are formed in addition to a sigma bond and are present in multiple bonds (double or triple). Advantages of VBT Explains bond characteristics (length, strength). Quantitative relationship between overlapping and bond dissociation energy. Accounts for molecular shape and bonding where covalency is not based on ground state half-filled orbitals. Redefined stability of molecules (e.g., BF$_3$, AlCl$_3$, PCl$_5$, SF$_6$). Disadvantages of VBT Predicts 90° bond angles for CH$_4$, NH$_3$, H$_2$O (based on p-p overlapping), but actual angles are 109°28', 107°, 104.5° respectively. Pauling introduced hybridization to explain characteristic geometrical shapes. Hybridisation Introduced by Pauling to explain equivalent nature of covalent bonds. Definition: Mixing of different shapes and approximately equal energy atomic orbitals to form new orbitals of same shape & energy (hybrid orbitals). Central atom undergoes hybridization. Process occurs before bond formation. Filled, half-filled, and empty orbitals can participate. Number of hybrid orbitals formed equals number of participating atomic orbitals. Each hybrid orbital has two lobes (major and minor); bond forms from major lobe. Hybrid orbitals have greater directional properties than atomic orbitals, forming stronger sigma bonds. Hybrid orbitals are arranged to minimize repulsion. Example: CH$_4$ C: [He] $2s^2 2p^2$ ($\uparrow\downarrow \quad \uparrow\uparrow\cdot$) C$^*$: [He] $2s^1 2p^3$ ($\uparrow \quad \uparrow\uparrow\uparrow$) Forms four $sp^3$ hybrid orbitals, leading to a tetrahedral structure. Determination of Hybridisation State Steric Number (S.N.) = Number of $\sigma$-bonds around atom + Number of lone pairs on atom. Molecule Steric Number Hybridisation NH$_3$ 4 ($\sigma$) + 0 (lp) = 4 $sp^3$ SF$_4$ 4 ($\sigma$) + 1 (lp) = 5 $sp^3d$ SO$_4^{2-}$ 4 ($\sigma$) + 0 (lp) = 4 $sp^3$ NO$_3^-$ 3 ($\sigma$) + 0 (lp) = 3 $sp^2$ Number of Hybrid Orbitals Hybridisation Two $sp$ Three $sp^2$ Four $sp^3$ Five $sp^3d$ Six $sp^3d^2$ Seven $sp^3d^3$ Types of Hybridisation (I) $sp$ Hybridisation One s-orbital & one p-orbital mix to form two new $sp$ hybrid orbitals. Arranged in a straight line at 180°. Example: Formation of BeF$_2$ molecule. Be (Ground State): $2s^2 2p^0$ ($\uparrow\downarrow$) Be (Excited State): $2s^1 2p^1$ ($\uparrow \quad \uparrow$) Forms two $sp$ hybrid orbitals. Examples: BeH$_2$, BeF$_2$, CO$_2$, C$_2$H$_2$, HCN. (II) $sp^2$ Hybridisation One s-orbital & two p-orbitals mix to form three new $sp^2$ hybrid orbitals. Arranged in a trigonal planar shape at 120°. Example: Formation of BF$_3$ molecule. B (Ground State): $2s^2 2p^1$ ($\uparrow\downarrow \quad \uparrow$) B (Excited State): $2s^1 2p^2$ ($\uparrow \quad \uparrow\uparrow$) Forms three $sp^2$ hybrid orbitals. Examples: BF$_3$, BCl$_3$, C$_2$H$_4$, Graphite, HNO$_3$. (III) $sp^3$ Hybridisation One s-orbital & three p-orbitals mix to form four new $sp^3$ hybrid orbitals. Angle between orbitals is 109°28' (tetrahedral). Example: Formation of Methane molecule. C (Ground State): $2s^2 2p^2$ ($\uparrow\downarrow \quad \uparrow\uparrow$) C (Excited State): $2s^1 2p^3$ ($\uparrow \quad \uparrow\uparrow\uparrow$) Forms four $sp^3$ hybrid orbitals. Examples: CH$_4$, CCl$_4$, NH$_3$, H$_2$O, Diamond. (IV) $sp^3d$ Hybridisation One s, three p, and one d ($d_{z^2}$) orbital mix to form five $sp^3d$ hybrid orbitals. Three orbitals at 120° in a trigonal plane, two perpendicular (trigonal bipyramidal shape). Example: PCl$_5$. P (Ground State): $3s^2 3p^3$ ($\uparrow\downarrow \quad \uparrow\uparrow\uparrow$) P (Excited State): $3s^1 3p^3 3d^1$ ($\uparrow \quad \uparrow\uparrow\uparrow \quad \uparrow$) Forms five $sp^3d$ hybrid orbitals. Axial P-Cl bonds are longer than equatorial P-Cl bonds due to repulsion. Examples: PF$_5$, PCl$_5$, SF$_4$, ClF$_3$. (V) $sp^3d^2$ Hybridisation One s, three p, and two d ($d_{z^2}$, $d_{x^2-y^2}$) orbitals mix to form six $sp^3d^2$ hybrid orbitals. All hybrid orbitals at 90° (octahedral shape). Example: SF$_6$. S (Ground State): $3s^2 3p^4$ ($\uparrow\downarrow \quad \uparrow\downarrow\uparrow\uparrow$) S (Excited State): $3s^1 3p^3 3d^2$ ($\uparrow \quad \uparrow\uparrow\uparrow \quad \uparrow\uparrow$) Forms six $sp^3d^2$ hybrid orbitals. Examples: SF$_6$, IF$_5$, XeOF$_4$. (VI) $sp^3d^3$ Hybridisation One s, three p, and three d ($d_{xy}$, $d_{xz}$, $d_{y^2-y^2}$) orbitals mix to form seven $sp^3d^3$ hybrid orbitals. Arranged in pentagonal bipyramidal shape. Five bond angles of 72°, two of 90°. Example: IF$_7$. I (Ground State): $5s^2 5p^5$ ($\uparrow\downarrow \quad \uparrow\downarrow\uparrow\downarrow\uparrow$) I (Excited State): $5s^1 5p^3 5d^3$ ($\uparrow \quad \uparrow\uparrow\uparrow \quad \uparrow\uparrow\uparrow$) Forms seven $sp^3d^3$ hybrid orbitals. Examples: IF$_7$, XeF$_6$. Atomic Orbitals Involved in Hybridisation $sp$: s + any p orbital $sp^2$: s + any two p orbitals $sp^3$: s + all p orbitals $sp^3d$: s + $p_x + p_y + p_z + d_{z^2}$ $sp^3d^2$: s + $p_x + p_y + p_z + d_{z^2} + d_{x^2-y^2}$ $sp^3d^3$: s + $p_x + p_y + p_z + d_{z^2} + d_{x^2-y^2} + d_{xy}$ Valence Shell Electron Pair Repulsion (VSEPR) Theory Explains molecular geometry based on repulsion between electron pairs in valence shell. Predicts molecular shape by minimizing electron-electron repulsion. Order of Repulsion: Lone pair-lone pair $>$ lone pair-bond pair $>$ bond pair-bond pair. Actual shapes of molecules with lone pairs are distorted (e.g., NH$_3$, H$_2$O bond angles). Shapes of Molecules Based on VSEPR Theory Total Hybrid Orbitals No. of B.P. No. of L.P. General Formula Hybridisation Stereochemical Formula/Structure Shape Example 2 2 0 AB$_2$ $sp$ B$-$A$-$B Linear BeCl$_2$ 3 3 0 AB$_3$ $sp^2$ Trigonal Planar BCl$_3$, NO$_3^-$ 3 2 1 AB$_2$ $sp^2$ Bent or Angular SnCl$_2$, O$_3$ 4 4 0 AB$_4$ $sp^3$ Tetrahedron CH$_4$, SiF$_4$ 4 3 1 AB$_3$ $sp^3$ Trigonal Pyramidal NH$_3$, CH$_3^-$ 4 2 2 AB$_2$ $sp^3$ V or Bent or Angular H$_2$O, SF$_2$ 4 1 3 AB $sp^3$ Linear ClO$^-$ 5 5 0 AB$_5$ $sp^3d$ Trigonal Bipyramidal PF$_5$, SF$_5$ 5 4 1 AB$_4$ $sp^3d$ Seesaw SF$_4$ 5 3 2 AB$_3$ $sp^3d$ T-shaped ClF$_3$, BrF$_3$ 5 2 3 AB$_2$ $sp^3d$ Linear ICl$_2^-$, XeF$_2$ 6 6 0 AB$_6$ $sp^3d^2$ Octahedral SF$_6$, IF$_6^+$ 6 5 1 AB$_5$ $sp^3d^2$ Square Pyramidal IF$_5$, XeOF$_4$ 6 4 2 AB$_4$ $sp^3d^2$ Square Planar XeF$_4$, ICl$_4^-$ 7 7 0 AB$_7$ $sp^3d^3$ Pentagonal Bipyramidal IF$_7$ 7 5 2 AB$_5$ $sp^3d^3$ Pentagonal Planar XeF$_5^-$ Planarity of Molecules To identify planarity, first predict hybridization and draw shape. In planar structures, all atoms are in the same plane; number of such planes is one. In non-planar structures, visualize in 3D then identify maximum atoms in a plane and count planes. Example: Number of planar SF$_4$ (1), CO$_3^{2-}$ (1), XeO$_3$F$_2$ (4). Bond Parameters (A) Bond Angle Angle between any two adjacent bonds. Represented in degrees (°), minutes ('), seconds ("). Factors Affecting Bond Angle Hybridisation or % 's' character: Bond angle $\propto$ %s character. BeCl$_2$ ($sp$, 180°) $>$ BCl$_3$ ($sp^2$, 120°) $>$ CCl$_4$ ($sp^3$, 109°28'). Lone Pair: When hybridisation is same, lone pairs affect bond angle. Bond angle $\propto \frac{1}{\text{No. of lone pair}}$. CH$_4$ ($sp^3$, 0 lp, 109°28') $>$ NH$_3$ ($sp^3$, 1 lp, 107°) $>$ H$_2$O ($sp^3$, 2 lp, 104.5°). If central atom has same number of lone pairs, bond angle depends on electronegativity of A & B. Electronegativity: Bond angle $\propto$ Electronegativity of central atom. If side atoms are same, increasing EN of central atom increases bond angle. Example: H$_2$O (EN of O $>$ S) $>$ H$_2$S. Bond angle of NH$_3$ $>$ PH$_3$ $>$ AsH$_3$. (EN of central atom decreasing, bond angle decreases). Side Atom: Bond angle $\propto \frac{1}{\text{size of side atom}}$. In AB$_x$ type molecules, if central atoms are same and EN of side atoms increases, bond angle decreases. PF$_3$ $ OF$_2$ $ Bond angle depends on size of side atom, increases with increasing size (Cl$_2$O $>$ H$_2$O). (B) Bond Length (Bond distance) Average distance between nuclei of two atoms. Represented in Å. Depends mainly on electronegativities of constituent atoms. Case I: If electronegativity difference is zero: Bond length = $r_A + r_B$. If $r_A = r_B$, Bond length = $2r_A$. (e.g., Cl$-$Cl). Case II: If electronegativity difference is not zero: Bond length = $r_A + r_B - 0.09 (\chi_A - \chi_B)$ (Schomaker and Stevenson formula). Factors Affecting Bond Length Electronegativity: Bond length $\propto \frac{1}{\Delta EN}$. H$-$F $ Bond Order: Bond length $\propto \frac{1}{\text{Number of bonds}}$. C$-$C (1.54 Å) $>$ C$=$C (1.34 Å) $>$ C$\equiv$C (1.20 Å). (C) Bond Energy (BE) Bond Formation Energy: Energy released when a bond is formed. Bond Dissociation Energy: Energy required to dissociate a bond. Calculated using dissociation energy. In diatomic molecules, Bond energy = bond dissociation energy. Example: N$_2$ $>$ O$_2$ $>$ H$_2$ $>$ F$_2$. Factors Affecting Bond Energy Electronegativity: Bond energy $\propto \Delta EN$. HF $>$ HCl $>$ HBr $>$ HI. Bond Order: Bond energy $\propto$ Bond order. C$-$C (79 K.Cal) $ Atomic Size: Bond energy $\propto \frac{1}{\text{Atomic size}}$. C$\equiv$C $>$ C$\equiv$N $>$ N$\equiv$N. Exception: Halogen group: Cl$-$Cl $>$ Br$-$Br $>$ F$-$F $>$ I$-$I. (F-F bond weakens due to electron repulsion). Bond Polarity: Bond energy $\propto$ Bond polarity. H$-$F $>$ H$-$Cl $>$ H$-$Br $>$ H$-$I. Resonance: Bond energy increases due to resonance. In benzene, bond energy of C$-$C increases due to $\pi$ electrons of C$=$C. Hybridisation: Bond energy $\propto$ s-character in hybrid orbitals. $sp-sp$ (50% s) $>$ $sp^2-sp^2$ (33.3% s) $>$ $sp^3-sp^3$ (25% s). Lone Pair of Electrons: Bond energy $\propto \frac{1}{\text{lone pair of electrons}}$. C$-$C $>$ N$-$N $>$ O$-$O $>$ F$-$F (for small atoms, lone pair repulsion weakens bond). Bent's Rule A lone pair of electrons prefers to occupy hybrid orbitals with greater % s-character. More electronegative atoms/groups prefer to occupy hybrid orbitals with smaller % s-character. S-orbital is closer to nucleus, so electron pairs in s-orbital experience greater attraction, increasing stability. More electronegative atoms attract shared electron pair towards themselves, so they prefer orbitals with less s-character to increase stability. Drago Generalisation For molecules meeting criteria: (i) 3rd or lower period, (ii) at least one lone pair, (iii) EN of surrounding atom $\le$ 2.5. Bond angle is approximately 90°, indicating no hybridization; overlapping occurs via pure atomic orbitals. Group 15: NH$_3$ (107°48') $>$ PH$_3$ (93°36') $>$ AsH$_3$ (91°48') $>$ SbH$_3$ (91°18'). Group 16: H$_2$O (104°28') $>$ H$_2$S (92°) $>$ H$_2$Se (91°) $>$ H$_2$Te (90.5°). Odd Electronic Species (1) NO$_2$ N-O bond length is between single and double bond length. Free electron occupies one $sp^2$ hybrid orbital. Paramagnetic (unpaired electron) brown gas. When cooled to -11°C, dimerizes to N$_2$O$_4$, which is diamagnetic (no unpaired electrons) white solid. Molecules That Do Not Exist SF$_4$, SF$_6$, PF$_5$ exist but OF$_4$, OF$_6$, NF$_5$ do not. Reason: N and O lack vacant 2d-orbitals to expand octet. PI$_5$(vap) & SCl$_6$ do not exist. Reason: Steric crowding of surrounding atoms. SCl$_6$ does not exist while TeCl$_6$ exists (size of Te $>$ S). PI$_5$(Solid) exists (steric crowding avoided by [PI$_4$]$^+$[I]$^-$ formation). SF$_6$, PF$_5$, XeF$_6$, XeF$_4$, XeF$_2$ exist but SH$_6$, PH$_5$, XeH$_6$, XeH$_4$, XeH$_2$ do not exist. Reason: F is more electronegative, causing d-orbital contraction, allowing hybridization. H is less electronegative, cannot cause d-orbital contraction, so required hybridization is not possible. Dipole Moment ($\mu$) (Ionic Nature in Covalent Bond) Due to electronegativity difference, covalent bonds acquire partial polar character. Degree of polarity expressed by dipole moment. $\mu = q \times d$ $q$: charge on one atom ($10^{-10}$ esu or $10^{-19}$ Coulomb). $d$: distance between charged atoms ($10^{-8}$ cm). $1 \text{ Debye (D)} = 10^{-18} \text{ esu cm} = 3.33 \times 10^{-30} \text{ Cm}$. Important Notes on Dipole Moment Symmetrical molecules without lone pairs have $\mu = 0$ (e.g., CO$_2$, BeF$_2$, BF$_3$, CCl$_4$). Dipole moment is a vector quantity; individual dipole moments add vectorially. In H-X bond, H is positive end, X is more electronegative. In C-X bond, C is positive end, X is other than carbon. C-H bond dipoles vary with hybridization. Diatomic molecules with different atoms always have dipole moment. Increases with electronegativity difference. Application of Dipole Moment To Determine Polarity and Geometry of Molecule: If $\mu = 0$, compound is non-polar and symmetrical (e.g., CO$_2$, BF$_3$, CCl$_4$, CH$_4$). If $\mu \neq 0$, compound is polar and unsymmetrical (e.g., H$_2$O, SO$_2$, NH$_3$, CH$_3$Cl). To Calculate % Ionic Character: % Ionic character = $\frac{\text{Experimental value of } \mu}{\text{Theoretical value of } \mu} \times 100$. Some Orders of Dipole Moments HF $>$ H$_2$O $>$ NH$_3$ $>$ NF$_3$. CH$_3$Cl $>$ CH$_3$F $>$ CH$_3$Br $>$ CH$_3$I. HF $>$ H$_2$O $>$ SO$_2$ $>$ NH$_3$ $>$ PH$_3$ $ NO $>$ NO$_2$ $>$ NO$_3^-$. CH$_3$Cl $>$ CH$_2$Cl$_2$ $>$ CHCl$_3$ $>$ CCl$_4$. H$_2$O $>$ H$_2$S. BF$_3$ $ H$_2$O $ Inorganic Substances (Dipole Moment) Monoatomic (He, Ne) and diatomic homonuclear (H$_2$, Cl$_2$, N$_2$) molecules have zero dipole moment (symmetrical). Triatomic molecules: CO$_2$, CS$_2$, HgCl$_2$ (linear, $\mu=0$). H$_2$O, SO$_2$ (angular, $\mu \neq 0$). Tetratomic molecules: BCl$_3$, BF$_3$, CO$_3^{2-}$, NO$_3^-$ (flat, symmetrical, $\mu=0$). PCl$_3$, AsCl$_3$, NH$_3$, PH$_3$, AsH$_3$, H$_3$O$^+$ (trigonal pyramidal, $\mu \neq 0$). Organic Substances (Dipole Moment) Methane (CH$_4$) and CCl$_4$ have zero dipole moment (symmetrical tetrahedral). Benzene has zero dipole moment (symmetrical hexagonal structure). Detects cis- and trans-isomers (cis-dibromoethylene $\mu \neq 0$, trans-dibromoethylene $\mu = 0$). Aromatic compounds: Dipole moments vary with orientation of groups (e.g., ortho, meta, para products). Hydrogen Bonding Electrostatic attractive force between covalently bonded H atom of one molecule and an electronegative atom (F, O, N & sp hybrid C). Not formed in ionic compounds. Forms in polar covalent compounds (not non-polar). Weaker than covalent bond but stronger than Van der Waals forces. Also known as dipole-dipole attraction. Main Condition for H-Bonding H covalently bonded to highly electronegative element (F, O, N). Atomic size of electronegative element should be small. Decreasing atomic size: N $>$ O $>$ F. Decreasing EN: F $>$ O $>$ N. Types of Hydrogen Bonding Intermolecular H-Bond: H-bond between two or more molecules (same or different compounds). More association, stronger. Homo intermolecular: H-bond between molecules of same compound (e.g., H$_2$O, HF). Hetero intermolecular: H-bond between molecules of different compounds (e.g., alcohol and water). Intramolecular H-Bond: H-bond within the same molecule. H-bonded with electronegative element of a functional group and another electronegative element in nearest position. Mostly in organic compounds. Results in ring formation (chelation). Less association than intermolecular H-bond, so less strength. Examples: o-nitrophenol, salicylaldehyde, o-fluorophenol. Effect of H-bond on Physical Properties (A) Solubility Intermolecular H-bonding: Few non-polar organic compounds are soluble in water due to H-bonding (e.g., glucose, fructose). Ketone, ether, alkane are insoluble. Dimethyl ether is soluble in water, diethyl ether is partially soluble (bulky ethyl groups hinder H-bonding). Solubility order: CH$_3$OCH$_3$ $ Intramolecular H-bonding: Decreases solubility as it forms chelate, preventing H-bond with water. (B) Viscosity Viscosity $\propto$ extent of H-bonding $\propto$ OH groups. H-bond associates molecules, increasing viscosity. CH$_2$OHCHOHCH$_2$OH (Glycerol) $>$ CH$_2$OHCHOH (Glycol) $>$ CH$_3$OH. H$_2$O $>$ CH$_3$OH $>$ CH$_3$OCH$_3$. H$_3$PO$_4$ $>$ H$_2$SO$_4$ $>$ HNO$_3$. (C) Surface Tension Surface tension $\propto$ extent of H-bonding. (D) Melting Point and Boiling Point Due to intermolecular H-bond, M.P. & B.P. increase. H$_2$O $>$ CH$_3$OH $>$ CH$_3$OCH$_3$. VIA hydrides: H$_2$O $>$ TeH$_2$ $>$ SeH$_2$ $>$ H$_2$S. VIIA hydrides: HF $>$ HI $>$ HBr $>$ HCl. Sudden increase in B.P. of NH$_3$, H$_2$O, HF due to H-bonding. Intramolecular H-bonding causes ring formation, decreasing intermolecular forces (Van der Waals), so M.P. & B.P. are low. o-derivative $ (E) Volatility Volatility $\propto \frac{1}{\text{H-bonding strength}}$. Intramolecular H-bonding $>$ Intermolecular H-bonding (more volatile). (F) Molecular Weight Molecular wt. of CH$_3$COOH is double due to dimer formation via H-bonding. (G) Physical States H$_2$O is liquid, H$_2$S is gas. Water and Ice: Both have H-bonding. Density of ice is less than water (open cage-like crystal structure). Van der Waals Forces Weakest intermolecular forces, significant for physical properties. Non-directional, non-valence cohesive forces. Attractive forces independent of other molecules. Explains solid, liquid, gaseous states of many molecules and inert gases. Types of Van der Waals Forces Dipole-dipole attraction: Attraction between oppositely charged poles of two polar molecules (e.g., H$_2$O, H-F, NH$_3$). Weaker than ion-dipole attraction. Dipole-induced dipole attraction: Occurs in mixtures of polar and non-polar molecules. Polar molecule induces polarity in non-polar molecule (e.g., H$_2$O and Cl$_2$). Instantaneous dipole-induced dipole (London Dispersion Forces): Weak intermolecular forces in non-polar gaseous molecules. Exist at low temperatures (e.g., F$_2$, Cl$_2$, N$_2$, noble gases). Other Weak Interactions Ion-dipole attraction: Polar molecules attracted by ions. Negative pole attracted by cation, positive pole by anion. Observed in solvation (e.g., Na$^+$ and Cl$^-$ in water). Ion-induced dipole attraction: Non-polar molecules polarized by ions (e.g., Na$^+$ and Cl$_2$). Factors Affecting London Dispersion Force Size of Molecule. Polarizable Electrons. Molecular Mass. Energy Distance Relationship for Different Interactions Ionic Interaction: $\propto \frac{1}{r}$. Ion dipole interaction: $\propto \frac{1}{r^2}$. Dipole - Dipole interaction: $\propto \frac{1}{r^3}$ (for stationary polar molecule). Ion - induced dipole interaction: $\propto \frac{1}{r^4}$. Dipole - induced dipole interaction: $\propto \frac{1}{r^6}$. Instantaneous dipole – induced dipole interaction: $\propto \frac{1}{r^6}$. Molecular Orbital Theory (MOT) Developed by F. Hund and R.S. Mulliken (1932). Salient Features: Electrons in molecules are in molecular orbitals. Molecular orbitals formed by combining atomic orbitals of comparable energies and proper symmetry. An electron in atomic orbital influenced by one nucleus; in molecular orbital by two or more nuclei (polycentric). Number of molecular orbitals equals number of combining atomic orbitals. Two atomic orbitals form two molecular orbitals (bonding and anti-bonding). Bonding molecular orbital has lower energy and greater stability than anti-bonding. Electron probability distribution around nuclei in molecule by molecular orbital. Molecular orbitals filled according to Aufbau principle, Pauli Exclusion principle, and Hund's Rule. Formation of Molecular Orbitals: Linear Combination of Atomic Orbitals (LCAO) Wave functions ($\psi$) of atomic orbitals combine linearly. $\Psi_{MO} = \Psi_A + \Psi_B$ (Bonding M.O.) for constructive interference (waves in same phase). $\Psi^*_{MO} = \Psi_A - \Psi_B$ (Antibonding M.O.) for destructive interference (waves out of phase). Conditions for Combination of Atomic Orbitals Atomic orbitals must have same or nearly same energy. (e.g., 1s of one atom combines with 1s of another, not 2s). Atomic orbitals must have same symmetry about the molecular axis (conventionally z-axis). (e.g., $2p_z$ combines with $2p_z$, not $2p_x$ or $2p_y$). Atomic orbitals must overlap to maximum extent. Greater overlap $\rightarrow$ greater electron probability density. Types of Molecular Orbitals Sigma ($\sigma$) molecular orbitals are symmetrical around internuclear axis. Pi ($\pi$) molecular orbitals are not symmetrical. (1) s-s combination of orbitals Forms $\sigma_{1s}$ (bonding) and $\sigma^*_{1s}$ (antibonding). (2) p-p combination of orbitals (end-to-end overlap) Forms $\sigma_{2p_z}$ (bonding) and $\sigma^*_{2p_z}$ (antibonding). (3) p-p combination of orbitals (side-by-side overlap) Forms $\pi_{2p_x}$ / $\pi_{2p_y}$ (bonding) and $\pi^*_{2p_x}$ / $\pi^*_{2p_y}$ (antibonding). (4) s-p combination of orbitals Forms $\sigma_{2s2p_z}$ (bonding) and $\sigma^*_{2s2p_z}$ (antibonding). Note-1: Molecular Orbital Wave Functions Designated as $\Psi_{(g)}$ (gerade, even) and $\Psi_{(u)}$ (ungerade, odd). 'g' or 'u' refers to symmetry about the orbital center. If sign of lobes remains same after rotation about line perpendicular to axis, orbital is gerade; if sign changes, ungerade. Energy Level Diagram from MOs Molecular orbital energy diagram for up to N$_2$ (molecules with $\le$ 14 electrons) $\sigma_{1s} Molecular orbital energy diagram for O$_2$ and F$_2$ (molecules with $>$ 14 electrons) $\sigma_{1s} $\sigma^*, \pi^*$ = antibonding molecular orbital. $\sigma, \pi$ = bonding molecular orbital. $\sigma_{1s}$ and $\sigma^*_{1s}$ orbitals are represented as KK (closed K-shell structure). Rules of Filling up of Molecular Orbital with Electrons Aufbau Principle: Lower energy orbital filled first. Pauli Exclusion Principle: Max two electrons per orbital. Hund's Rule: If MOs have same energy, fill singly first, then pair. Electronic Configuration And Molecular Behaviour Stability: Molecule is stable if $N_b > N_a$. Unstable if $N_b \le N_a$. $N_b$: number of electrons in bonding MOs. $N_a$: number of electrons in antibonding MOs. Bond Order (B.O.) B.O. $= \frac{1}{2} (N_b - N_a)$. Positive B.O. ($N_b > N_a$) means stable molecule. Negative or zero B.O. means unstable molecule. Integral B.O. values (1, 2, 3) correspond to single, double, triple bonds. Bond-Length B.O. is approximate measure of bond length. Bond length decreases as B.O. increases. Magnetic Nature If one or more molecular orbitals are singly occupied, it is paramagnetic (attracted by magnetic field) (e.g., O$_2$). Otherwise diamagnetic (e.g., N$_2$). Molecular Orbital Diagram in Some Homonuclear Diatomic Species Species MO Configuration N$_b$ N$_a$ B.O. Magnetic Behaviour H$_2$ $(\sigma_{1s})^2$ 2 0 1 Diamagnetic He$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2$ 2 2 0 Li$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2$ 4 2 1 Diamagnetic Be$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2$ 4 4 0 B$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2p_x}^1 = \pi_{2p_y}^1)$ 6 4 1 Paramagnetic C$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2)$ 8 4 2 Diamagnetic N$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\sigma_{2p_z})^2$ 10 4 3 Diamagnetic N$_2^+$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\sigma_{2p_z})^1$ 9 4 2.5 Paramagnetic N$_2^-$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\sigma_{2p_z})^2 (\pi^*_{2p_x})^1$ 10 5 2.5 Paramagnetic O$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\pi^*_{2p_x}^1 = \pi^*_{2p_y}^1)$ 10 6 2 Paramagnetic O$_2^+$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\pi^*_{2p_x})^1$ 10 5 2.5 Paramagnetic O$_2^{2+}$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2)$ 10 4 3 Diamagnetic O$_2^-$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\pi^*_{2p_x}^2 = \pi^*_{2p_y}^1)$ 10 7 1.5 Paramagnetic O$_2^{2-}$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\pi^*_{2p_x}^2 = \pi^*_{2p_y}^2)$ 10 8 1 Diamagnetic F$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\pi^*_{2p_x}^2 = \pi^*_{2p_y}^2)$ 10 8 1 Diamagnetic Ne$_2$ $(\sigma_{1s})^2 (\sigma^*_{1s})^2 (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_{2p_x}^2 = \pi_{2p_y}^2) (\pi^*_{2p_x}^2 = \pi^*_{2p_y}^2) (\sigma^*_{2p_z})^2$ 10 10 0 Colour of Halogens F$_2$ (yellow), Cl$_2$ (greenish yellow), Br$_2$ (reddish brown), I$_2$ (violet). Reason: HOMO-LUMO transition. Energy gap decreases down the group, leading to different colours. Ionisation Energy Compare HOMO energy level with atomic orbital energy. If HOMO has high energy, electron removal is easier. Hydration Energy Energy released when 1 mole of gaseous ions are hydrated in water. Directly proportional to nuclear charge, inversely proportional to size. Decreases down the group. As charge density increases, hydrated size (aqueous radius) increases. Li$^+$(aq) $>$ Na$^+$(aq) $>$ K$^+$(aq) $>$ Rb$^+$(aq) $>$ Cs$^+$(aq). As hydrated size increases, ionic mobility decreases, reducing conductivity. Li$^+$(aq) $ Hydration energy affects solubility of ionic compounds. If hydration energy $>$ lattice energy, soluble in water. Fajan's Rule Explains partial covalent character in ionic bonds. Rules: Smaller cation size, larger anion size $\rightarrow$ greater covalent character. Greater charge on cation $\rightarrow$ greater covalent character. Cations with $(n-1)d^{x}ns^0$ configuration (transition metals) are more polarizing than noble gas configuration ($ns^2np^6$). Cation polarizes anion, pulling electron charge towards itself, increasing electron density between them (covalent character). Polarization Power: Ability of cation to polarize a nearby anion. Usually called ionic potential or charge density. Ionic potential $\phi = \frac{\text{Charge on cation}}{\text{Size of cation}}$. Polarizability: Ability of anion to get polarized by cation. Polarization of anion leads to electron sharing, imparting covalent character. Increased polarization $\rightarrow$ increased covalent character. Factors Affecting Polarization (Fajan's Rule) Size of Cation: Polarization of anion increases as cation size decreases. In a group (BeCl$_2$ $>$ MgCl$_2$ $>$ CaCl$_2$ $>$ SrCl$_2$ $>$ BaCl$_2$), cation size increases, covalent character decreases, ionic character increases. Be$^{2+}$ has greatest polarizing power. In a period (Na$^+$, Mg$^{2+}$, Al$^{3+}$, Si$^{4+}$), cation size decreases, covalent character increases. Size of Anion: If anion size increases for a given cation, covalent character increases. CaF$_2$ $ Charge on Cation and Anion: Polarization increases with increase in charge. Charge on cation and anion $\propto$ Polarization (covalent character). NaCl $ Electronic Configuration of Cation: Cations with pseudo inert gas configuration have high polarization capacity. CuCl (covalent) vs NaCl (ionic). Cu$^+$ (pseudo-inert) has greater polarizing power than Na$^+$ (inert). Increased Polarization Effects Melting Point: Increased covalent character causes decreasing melting point. Solubility in Polar Solvents: Increased covalent character decreases solubility in polar solvents. AgF $>$ AgCl $>$ AgBr $>$ AgI (solubility decreases). HgF$_2$ $>$ HgCl$_2$ $>$ HgBr$_2$ $>$ HgI$_2$. Electrical Conductance in Fused State and Tendency to Ionize in Solution: Increased covalent character decreases conductivity. Compound Conductance (ohm$^{-1}$ cm$^{-2}$) LiCl 166 BeCl$_2$ 0.086 NaCl 133 MgCl$_2$ 29 Intensity of Colour: Polarized anions cause colour. Intensity increases as polarization increases (Cl to I). HgCl$_2$ (colorless) $\rightarrow$ HgI$_2$ (red). AgCl (colorless) $\rightarrow$ AgI (yellow). SnCl$_4$ (colorless) $\rightarrow$ SnI$_4$ (red). PbCl$_2$ (colorless) $\rightarrow$ PbI$_2$ (dark yellow).