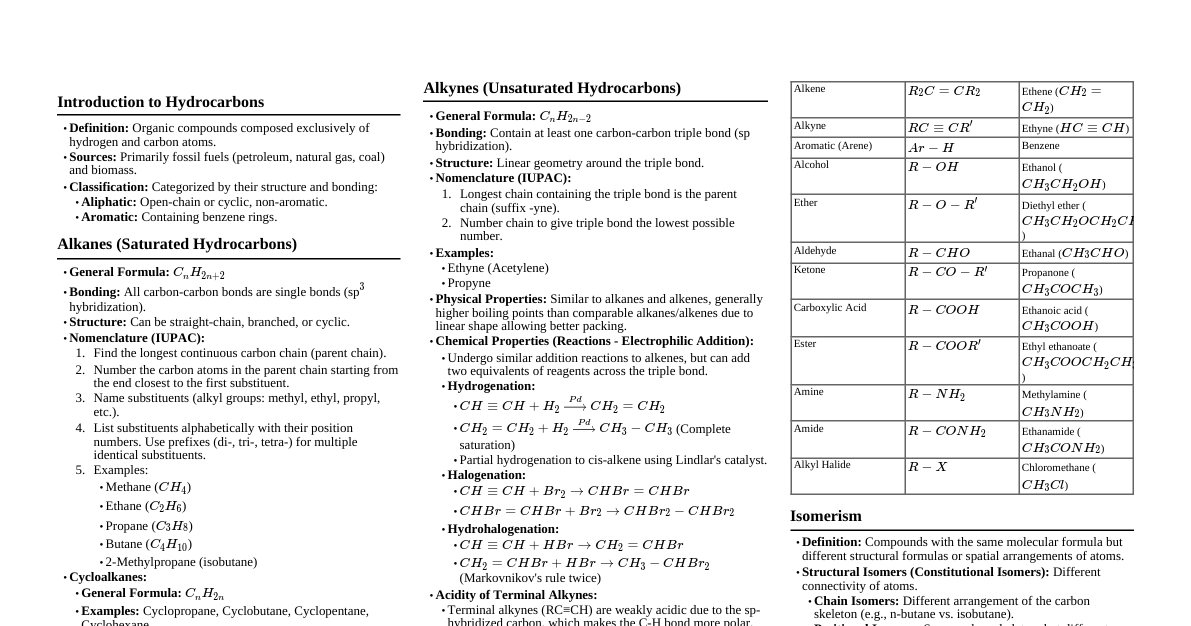
### General Organic Chemistry (GOC) #### 1. Electronic Effects * **Inductive Effect (I-effect):** * Permanent displacement of $\sigma$-electrons towards more electronegative atom. * **+I groups:** Alkyl groups (electron donating, e.g., $-\text{CH}_3$, $-\text{C}_2\text{H}_5$). Increases electron density. * **-I groups:** Halogens, $-\text{NO}_2$, $-\text{CN}$, $-\text{COOH}$, $-\text{OR}$ (electron withdrawing). Decreases electron density. * Effect decreases rapidly with distance. * **Application:** Influences acidity/basicity, stability of carbocations/carbanions. * Acidity: $-\text{COOH}$ with $-\text{I}$ groups is more acidic. * Basicity: Amines with $+\text{I}$ groups are more basic (in gas phase/aprotic solvent). * **Resonance Effect (Mesomeric Effect, M-effect):** * Delocalization of $\pi$-electrons or lone pairs. * **+M groups:** Groups with lone pairs (e.g., $-\text{OH}$, $-\text{OR}$, $-\text{NH}_2$, Halogens). Donates electrons to conjugated system. * **-M groups:** Groups with multiple bonds where the atom connected to the system is more electronegative (e.g., $-\text{NO}_2$, $-\text{CHO}$, $-\text{COOH}$, $-\text{CN}$). Withdraws electrons from conjugated system. * Effect is stronger than inductive effect. * **Resonance Structures:** Represent actual molecule as a hybrid. More stable resonance structures (more covalent bonds, no charge separation, negative charge on more electronegative atom) contribute more. * **Hyperconjugation (No-bond Resonance):** * Delocalization of $\sigma$-electrons of C-H bond of an alkyl group directly attached to an unsaturated system (double bond, triple bond, carbocation, or free radical). * **Effect:** Stabilizes carbocations, free radicals, and alkenes. More $\alpha$-hydrogens, more hyperconjugation, more stability. * **Stability Order:** Carbocation: $3^\circ > 2^\circ > 1^\circ > \text{methyl}$ (due to hyperconjugation and +I). Alkene: More substituted alkene is more stable. #### 2. Acid-Base Concepts * **Acidity:** Ability to donate a proton ($\text{H}^+$). * Factors increasing acidity: * Stable conjugate base (e.g., due to resonance, -I effect). * Increased s-character (e.g., $\text{sp}$ C-H is more acidic than $\text{sp}^2$ or $\text{sp}^3$). * Electronegativity of atom bearing the negative charge. * **Order:** $\text{CH}\equiv\text{CH} > \text{NH}_3 > \text{CH}_2=\text{CH}_2 > \text{CH}_3-\text{CH}_3$. * Carboxylic acids are more acidic than phenols due to more stable resonance structures of carboxylate ion. Phenols are more acidic than alcohols. * **Basicity:** Ability to accept a proton or donate a lone pair. * Factors increasing basicity: * Availability of lone pair (e.g., +I effect, no resonance delocalization of lone pair). * Less electronegative atom bearing the lone pair. * **Order for Amines (gas phase):** $3^\circ > 2^\circ > 1^\circ > \text{NH}_3$ (due to +I effect of alkyl groups). * **Order for Amines (aqueous phase):** $2^\circ > 3^\circ > 1^\circ > \text{NH}_3$ (due to solvation effects and steric hindrance alongside +I effect). * Aromatic amines are less basic than aliphatic amines due to resonance delocalization of lone pair on nitrogen into the benzene ring. **JEE Advanced Trick: Aromaticity** * **Huckel's Rule:** Planar, cyclic, fully conjugated system with $(4n+2)\pi$ electrons ($n=0, 1, 2, ...$). * **Anti-aromatic:** Planar, cyclic, fully conjugated with $(4n)\pi$ electrons. Highly unstable. * **Non-aromatic:** Any system not meeting criteria (e.g., non-planar, not fully conjugated). #### 3. Isomerism * **Structural Isomerism:** Same molecular formula, different connectivity. * **Chain Isomerism:** Different carbon skeleton (e.g., n-butane vs isobutane). * **Position Isomerism:** Different position of functional group/substituent (e.g., 1-propanol vs 2-propanol). * **Functional Group Isomerism:** Different functional group (e.g., ethanol vs dimethyl ether). * **Metamerism:** Different alkyl groups around a polyvalent functional group (e.g., diethyl ether vs methyl propyl ether). * **Tautomerism:** Special type of functional group isomerism, involving rapid interconversion of two isomers (tautomeres) by migration of an atom (usually H) and a $\pi$-bond (e.g., keto-enol tautomerism). Enol form is usually less stable except for $\beta$-dicarbonyl compounds or phenols. * **Stereoisomerism:** Same molecular formula, same connectivity, different spatial arrangement. * **Conformational Isomerism:** Interconvertible by rotation around C-C single bond (e.g., staggered, eclipsed for ethane; chair, boat for cyclohexane). Staggered/anti are more stable. * **Configurational Isomerism:** Requires bond breaking to interconvert. * **Geometric Isomerism (cis-trans):** Restricted rotation around C=C bond (or in cyclic compounds). Requires two different groups on each sp2 carbon. *E/Z notation* for more complex cases. * **Optical Isomerism:** Non-superimposable mirror images (enantiomers). Requires chiral center (carbon with four different groups). * **Chiral Molecule:** Optically active. Rotates plane-polarized light. * **Achiral Molecule:** Optically inactive. * **Diastereomers:** Stereoisomers that are not mirror images. * **Meso Compound:** Contains chiral centers but is achiral due to internal plane of symmetry. Optically inactive. * **Racemic Mixture:** Equimolar mixture of enantiomers. Optically inactive (external compensation). **JEE Mains Trap: Identifying Isomers** * Always check molecular formula first. * For geometric isomers, ensure each doubly bonded carbon has two different groups. * For optical isomers, look for chiral centers. Remember meso compounds are achiral despite chiral centers. ### Hydrocarbons: Alkanes #### 1. Preparation * **Hydrogenation of Alkenes/Alkynes:** $\text{R-CH=CH-R'} + \text{H}_2 \xrightarrow{\text{Ni/Pt/Pd}} \text{R-CH}_2-\text{CH}_2-\text{R}'$. * **Wurtz Reaction:** $2\text{RX} + 2\text{Na} \xrightarrow{\text{Dry ether}} \text{R-R} + 2\text{NaX}$. * Only for symmetrical alkanes. For unsymmetrical, mixture of products. * Cannot prepare methane. * **Decarboxylation of Carboxylic Acids:** $\text{R-COONa} + \text{NaOH} \xrightarrow{\text{CaO, Heat}} \text{R-H} + \text{Na}_2\text{CO}_3$. * Produces alkane with one carbon less than starting acid. * **Kolbe's Electrolytic Method:** $2\text{R-COONa} + 2\text{H}_2\text{O} \xrightarrow{\text{Electrolysis}} \text{R-R} + 2\text{CO}_2 + \text{H}_2 + 2\text{NaOH}$. * Forms symmetrical alkanes. Free radical mechanism. #### 2. Reactions * **Halogenation (Free Radical Substitution):** $\text{CH}_4 + \text{Cl}_2 \xrightarrow{\text{hv or Heat}} \text{CH}_3\text{Cl} + \text{HCl}$. * Chain reaction: Initiation, Propagation, Termination. * **Reactivity Order:** $\text{F}_2 > \text{Cl}_2 > \text{Br}_2 > \text{I}_2$. * **Selectivity Order:** $\text{I}_2 > \text{Br}_2 > \text{Cl}_2 > \text{F}_2$. (Bromination is more selective, prefers $3^\circ$ H removal). * **Stability of Free Radicals:** $3^\circ > 2^\circ > 1^\circ > \text{methyl}$. This determines product distribution. * **Combustion:** $\text{C}_n\text{H}_{2n+2} + (\frac{3n+1}{2})\text{O}_2 \rightarrow n\text{CO}_2 + (n+1)\text{H}_2\text{O} + \text{Heat}$. * **Isomerization:** $\text{n-alkane} \xrightarrow{\text{Anhydrous AlCl}_3/\text{HCl, Heat}} \text{branched alkane}$. ### Hydrocarbons: Alkenes #### 1. Preparation * **Dehydration of Alcohols:** $\text{R-CH}_2-\text{CH}_2-\text{OH} \xrightarrow{\text{Conc. H}_2\text{SO}_4/\text{Al}_2\text{O}_3, \text{Heat}} \text{R-CH=CH}_2 + \text{H}_2\text{O}$. * Follows Zaitsev's Rule (more substituted alkene is major product). Carbocation intermediate, rearrangements possible. * **Dehydrohalogenation of Alkyl Halides:** $\text{R-CH}_2-\text{CH}_2-\text{X} + \text{KOH(alc.)} \rightarrow \text{R-CH=CH}_2 + \text{KX} + \text{H}_2\text{O}$. * Follows Zaitsev's Rule. E2 mechanism. * **Dehalogenation of Vicinal Dihalides:** $\text{R-CHBr-CHBr-R'} + \text{Zn} \rightarrow \text{R-CH=CH-R'} + \text{ZnBr}_2$. #### 2. Reactions (Electrophilic Addition) * **Addition of H$_2$ (Hydrogenation):** $\text{R-CH=CH-R'} + \text{H}_2 \xrightarrow{\text{Ni/Pt/Pd}} \text{R-CH}_2-\text{CH}_2-\text{R}'$. (Syn addition). * **Addition of X$_2$ (Halogenation):** $\text{R-CH=CH-R'} + \text{Br}_2 \xrightarrow{\text{CCl}_4} \text{R-CHBr-CHBr-R}'$. (Anti addition). * Used as a test for unsaturation (decolorizes bromine water). * **Addition of HX (Hydrohalogenation):** $\text{R-CH=CH}_2 + \text{HX} \rightarrow \text{R-CHX-CH}_3$. * **Markovnikov's Rule:** Negative part of addendum goes to carbon with fewer hydrogens. Carbocation intermediate, rearrangements possible. * **Anti-Markovnikov's Rule (Peroxide Effect):** Only for HBr in presence of peroxides. Free radical mechanism. * **Addition of H$_2$O (Hydration):** $\text{R-CH=CH}_2 + \text{H}_2\text{O} \xrightarrow{\text{H}^+} \text{R-CH(OH)-CH}_3$. * Markovnikov's addition. Carbocation intermediate. * **Oxymercuration-Demercuration:** 1. $\text{R-CH=CH}_2 + \text{Hg(OAc)}_2, \text{H}_2\text{O}$ 2. $\text{NaBH}_4$ $\rightarrow \text{R-CH(OH)-CH}_3$. Markovnikov's addition of H and OH, no rearrangements. * **Hydroboration-Oxidation:** 1. $\text{R-CH=CH}_2 + \text{BH}_3 \cdot \text{THF}$ 2. $\text{H}_2\text{O}_2, \text{OH}^-$ $\rightarrow \text{R-CH}_2-\text{CH}_2-\text{OH}$. Anti-Markovnikov's addition of H and OH, Syn addition. * **Ozonolysis:** * **Reductive:** $\text{R-CH=CH-R'} + \text{O}_3 \xrightarrow{\text{Zn/H}_2\text{O}} \text{R-CHO} + \text{R'-CHO}$. Cleaves double bond, forms aldehydes/ketones. * **Oxidative:** $\text{R-CH=CH-R'} + \text{O}_3 \xrightarrow{\text{H}_2\text{O}_2} \text{R-COOH} + \text{R'-COOH}$. Forms carboxylic acids (or ketones if R is not H). * **Baeyer's Reagent (Cold, Dilute, Alkaline KMnO$_4$):** $\text{R-CH=CH-R'} \xrightarrow{\text{KMnO}_4/\text{OH}^-/\text{Cold}} \text{R-CH(OH)-CH(OH)-R}'$. Forms vicinal diols (syn addition). Decolorizes KMnO$_4$ (test for unsaturation). * **Hot, Acidified KMnO$_4$:** Cleaves double bond, similar to oxidative ozonolysis, forms carboxylic acids/ketones. **JEE Advanced Tip: Rearrangements** * Always check for carbocation rearrangements (hydride or alkyl shifts) in reactions involving carbocation intermediates (e.g., acid-catalyzed hydration, $\text{HX}$ addition, alcohol dehydration). This leads to more stable carbocations. ### Hydrocarbons: Alkynes #### 1. Preparation * **From Vicinal Dihalides:** $\text{R-CHX-CHX-R'} + 2\text{KOH(alc.)} \rightarrow \text{R-C}\equiv\text{C-R}' + 2\text{KX} + 2\text{H}_2\text{O}$. * **From Geminal Dihalides:** $\text{R-CX}_2-\text{CH}_2-\text{R}' + 2\text{NaNH}_2 \rightarrow \text{R-C}\equiv\text{C-R}' + 2\text{NaX} + 2\text{NH}_3$. * **From Calcium Carbide:** $\text{CaC}_2 + 2\text{H}_2\text{O} \rightarrow \text{CH}\equiv\text{CH} + \text{Ca(OH)}_2$. #### 2. Reactions (Electrophilic Addition) * **Addition of H$_2$ (Hydrogenation):** * **Complete:** $\text{R-C}\equiv\text{C-R'} + 2\text{H}_2 \xrightarrow{\text{Ni/Pt/Pd}} \text{R-CH}_2-\text{CH}_2-\text{R}'$. * **Partial (Syn):** $\text{R-C}\equiv\text{C-R'} + \text{H}_2 \xrightarrow{\text{Lindlar's Catalyst (Pd/CaCO}_3/\text{BaSO}_4)} \text{cis-R-CH=CH-R}'$. * **Partial (Anti):** $\text{R-C}\equiv\text{C-R'} + \text{Na}/\text{Li} \xrightarrow{\text{Liquid NH}_3} \text{trans-R-CH=CH-R}'$ (Birch reduction). * **Addition of X$_2$ (Halogenation):** $\text{R-C}\equiv\text{C-R'} + \text{X}_2 \rightarrow \text{R-CX=CX-R}' + \text{X}_2 \rightarrow \text{R-CX}_2-\text{CX}_2-\text{R}'$. * **Addition of HX (Hydrohalogenation):** $\text{R-C}\equiv\text{CH} + \text{HX} \rightarrow \text{R-CX=CH}_2 + \text{HX} \rightarrow \text{R-CX}_2-\text{CH}_3$. * Follows Markovnikov's rule twice. * **Addition of H$_2$O (Hydration):** $\text{R-C}\equiv\text{CH} + \text{H}_2\text{O} \xrightarrow{\text{HgSO}_4/\text{H}_2\text{SO}_4} \text{R-C(OH)=CH}_2 \rightleftharpoons \text{R-CO-CH}_3$. * Forms enol, which tautomerizes to ketone (Markovnikov's addition). * Ethyne gives ethanal ($\text{CH}_3\text{CHO}$). * **Acidity of Terminal Alkynes:** $\text{R-C}\equiv\text{CH} + \text{Na} \rightarrow \text{R-C}\equiv\text{C}^-\text{Na}^+ + \frac{1}{2}\text{H}_2$. * Due to $\text{sp}$ hybridized carbon, which is more electronegative, making the C-H bond slightly acidic. * Reacts with strong bases like $\text{NaNH}_2$ or metals. Does not react with $\text{NaOH}$ or $\text{KOH}$. * Forms silver mirror with Tollen's reagent ($\text{Ag(NH}_3)_2^+$). * Forms red precipitate with ammoniacal cuprous chloride ($\text{CuCl}$). ### Hydrocarbons: Aromatic Compounds (Benzene) #### 1. Preparation * **Cyclic Polymerization of Ethyne:** $3\text{CH}\equiv\text{CH} \xrightarrow{\text{Red hot iron tube, 873K}} \text{C}_6\text{H}_6$. * **Decarboxylation of Benzoic Acid:** $\text{C}_6\text{H}_5\text{COONa} + \text{NaOH} \xrightarrow{\text{CaO, Heat}} \text{C}_6\text{H}_6 + \text{Na}_2\text{CO}_3$. * **Reduction of Phenol:** $\text{C}_6\text{H}_5\text{OH} + \text{Zn dust} \xrightarrow{\text{Heat}} \text{C}_6\text{H}_6 + \text{ZnO}$. #### 2. Reactions (Electrophilic Aromatic Substitution, EAS) * **Nitration:** $\text{C}_6\text{H}_6 + \text{HNO}_3 \xrightarrow{\text{Conc. H}_2\text{SO}_4} \text{C}_6\text{H}_5\text{NO}_2 + \text{H}_2\text{O}$. * Electrophile: $\text{NO}_2^+$. * **Halogenation:** $\text{C}_6\text{H}_6 + \text{X}_2 \xrightarrow{\text{FeX}_3 \text{ or Anhydrous AlX}_3} \text{C}_6\text{H}_5\text{X} + \text{HX}$. * Electrophile: $\text{X}^+$. * **Sulfonation:** $\text{C}_6\text{H}_6 + \text{Conc. H}_2\text{SO}_4/\text{Fuming H}_2\text{SO}_4 \rightleftharpoons \text{C}_6\text{H}_5\text{SO}_3\text{H} + \text{H}_2\text{O}$. * Reversible. Electrophile: $\text{SO}_3$. * **Friedel-Crafts Alkylation:** $\text{C}_6\text{H}_6 + \text{R-Cl} \xrightarrow{\text{Anhydrous AlCl}_3} \text{C}_6\text{H}_5\text{R} + \text{HCl}$. * Electrophile: $\text{R}^+$. Carbocation rearrangements possible. * Polyalkylation can occur. * **Friedel-Crafts Acylation:** $\text{C}_6\text{H}_6 + \text{R-COCl} \xrightarrow{\text{Anhydrous AlCl}_3} \text{C}_6\text{H}_5\text{COR} + \text{HCl}$. * Electrophile: Acylium ion ($\text{R-C}\equiv\text{O}^+$). No rearrangements. * Prevents polyacylation as the product ketone is deactivating. #### 3. Directing Groups in EAS * **Ortho-Para Directors (+M, +I):** Activating. Increase electron density at o- and p-positions. * $-\text{NH}_2$, $-\text{NR}_2$, $-\text{OH}$, $-\text{OR}$, $-\text{NHCOR}$, $-\text{R}$, $-\text{Ar}$. * Halogens are o,p-directing but deactivating (due to strong -I effect outweighing +M effect). * **Meta Directors (-M, -I):** Deactivating. Decrease electron density on ring, especially at o- and p-positions. * $-\text{NO}_2$, $-\text{CN}$, $-\text{CHO}$, $-\text{COR}$, $-\text{COOH}$, $-\text{COOR}$, $-\text{SO}_3\text{H}$. **JEE Advanced Trap: Reactivity and Directing Effects** * For disubstituted benzenes, the directing effect of the stronger activating group usually dictates the position of the incoming electrophile. * Steric hindrance can also play a role, favoring para over ortho if both are activated. #### 4. Other Reactions * **Combustion:** Burns with sooty flame (high C:H ratio). * **Hydrogenation:** $\text{C}_6\text{H}_6 + 3\text{H}_2 \xrightarrow{\text{Ni, 473-573K}} \text{C}_6\text{H}_{12}$ (cyclohexane). * **Addition of Cl$_2$ (in presence of UV light):** $\text{C}_6\text{H}_6 + 3\text{Cl}_2 \xrightarrow{\text{hv}} \text{C}_6\text{H}_6\text{Cl}_6$ (Benzene Hexachloride, BHC or Gammaxene). This is free radical addition, not substitution. * **Oxidation:** Benzene is resistant to strong oxidizing agents like $\text{KMnO}_4$. However, side chains on benzene can be oxidized: * $\text{Toluene} \xrightarrow{\text{KMnO}_4/\text{KOH, Heat}} \text{Benzoic acid}$. Any alkyl group on benzene, regardless of length, is oxidized to a carboxylic acid group, provided it has at least one benzylic hydrogen.