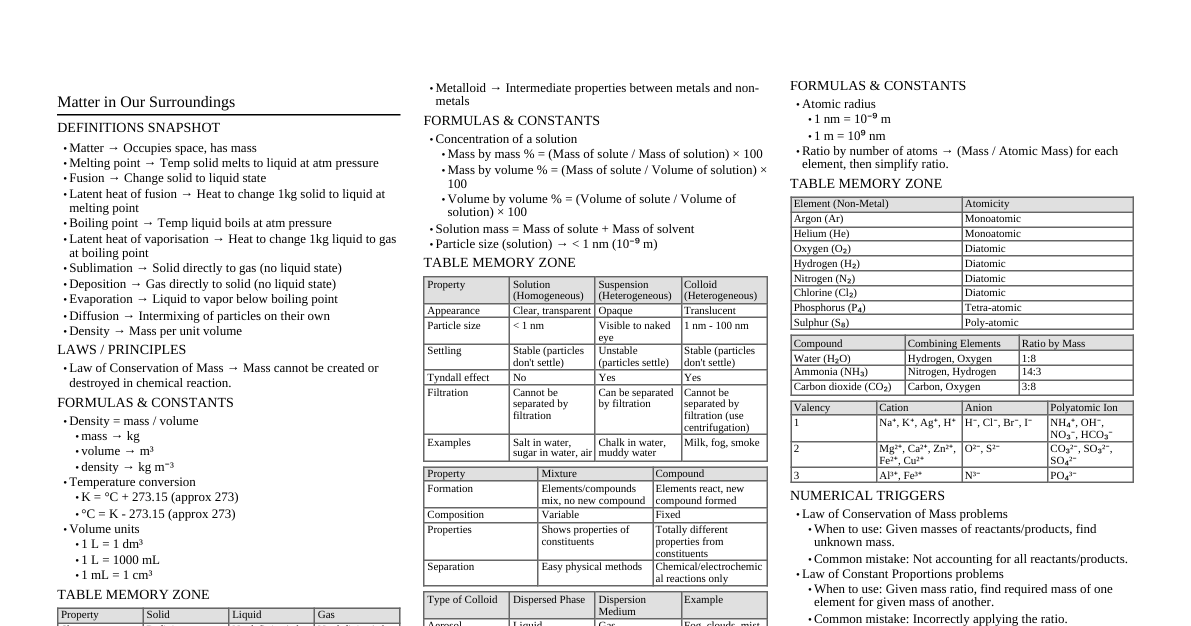
### Chemical Reactions and Equations A **chemical reaction** is a process that involves rearrangement of the molecular or ionic structure of a substance, as opposed to a change in physical form or a nuclear reaction. During a chemical reaction, atoms of one element do not change into those of another element. Only a rearrangement of atoms takes place. **Characteristics of Chemical Reactions:** 1. **Change in State:** Reactants can be solids, liquids, or gases, and products can have a different state. * *Example:* Burning of candle wax (solid) produces water (liquid) and carbon dioxide (gas). 2. **Change in Colour:** The colour of the reactants may change during the reaction. * *Example:* When citric acid reacts with purple potassium permanganate solution, the purple colour disappears. 3. **Evolution of Gas:** Some reactions produce gas as a product, which can be observed as bubbles. * *Example:* When zinc granules react with dilute sulphuric acid, hydrogen gas is evolved. 4. **Change in Temperature:** Reactions can either release heat (exothermic) or absorb heat (endothermic). * *Example (Exothermic):* Quicklime reacting with water to form slaked lime. * *Example (Endothermic):* Barium hydroxide reacting with ammonium chloride. 5. **Formation of a Precipitate:** An insoluble solid (precipitate) may form when two solutions are mixed. * *Example:* When potassium iodide solution is added to lead nitrate solution, a yellow precipitate of lead iodide is formed. ### Chemical Equations A **chemical equation** is a symbolic representation of a chemical reaction in the form of symbols and formulae, wherein the reactant entities are given on the left-hand side and the product entities on the right-hand side with a plus sign between the entities in both the reactants and the products and an arrow pointing towards the products to show the direction of the reaction. **Writing a Chemical Equation:** * Reactants are written on the left-hand side (LHS) with a plus sign (+) between them. * Products are written on the right-hand side (RHS) with a plus sign (+) between them. * An arrow ($\rightarrow$) separates reactants from products, indicating the direction of the reaction. * The physical states of reactants and products are indicated in parentheses: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution. *Example:* $$ \text{Magnesium (s) + Oxygen (g)} \rightarrow \text{Magnesium Oxide (s)} $$ $$ 2Mg(s) + O_2(g) \rightarrow 2MgO(s) $$ **Balanced Chemical Equation:** A **balanced chemical equation** is an equation in which the number of atoms of each element is the same on both the reactant side and the product side. This is based on the **Law of Conservation of Mass**, which states that mass can neither be created nor destroyed in a chemical reaction. *Example:* $$ Fe(s) + H_2O(g) \rightarrow Fe_3O_4(s) + H_2(g) $$ *Unbalanced.* To balance: $$ 3Fe(s) + 4H_2O(g) \rightarrow Fe_3O_4(s) + 4H_2(g) $$ *Balanced.* **Steps for Balancing Chemical Equations (Hit and Trial Method):** 1. Write the unbalanced equation with correct chemical formulae. 2. Count the number of atoms of each element on both sides. 3. Start balancing with the compound having the maximum number of atoms. It could be a reactant or a product. Pick the element in that compound which has the maximum number of atoms. 4. Balance the atoms of the selected element by multiplying the formula with a suitable coefficient. 5. Balance other elements one by one. 6. Check the equation for all elements. 7. Write the physical states of reactants and products. **Activity 1.1:** Burning of Magnesium Ribbon * **Observation:** A magnesium ribbon burns in air with a dazzling white flame and changes into a white powder (magnesium oxide). * **Equation:** $2Mg(s) + O_2(g) \rightarrow 2MgO(s)$ * **Note:** Magnesium ribbon should be cleaned with sandpaper before burning to remove the protective layer of magnesium oxide/carbonate, which prevents it from reacting readily. **Activity 1.2:** Reaction of Lead Nitrate and Potassium Iodide * **Observation:** When solutions of lead nitrate and potassium iodide are mixed, a yellow precipitate of lead iodide is formed. * **Equation:** $Pb(NO_3)_2(aq) + 2KI(aq) \rightarrow PbI_2(s) \downarrow + 2KNO_3(aq)$ * **Type:** Double Displacement Reaction (Precipitation Reaction). **Activity 1.3:** Reaction of Zinc with Dilute Sulphuric Acid * **Observation:** When zinc granules are added to dilute sulphuric acid, hydrogen gas is evolved, and the flask becomes hot. * **Equation:** $Zn(s) + H_2SO_4(aq) \rightarrow ZnSO_4(aq) + H_2(g) \uparrow$ * **Type:** Single Displacement Reaction, Exothermic Reaction. ### Types of Chemical Reactions #### 1. Combination Reaction A **combination reaction** is a reaction in which two or more reactants combine to form a single product. *General Form:* $A + B \rightarrow AB$ *Examples:* * **Burning of Coal:** $C(s) + O_2(g) \rightarrow CO_2(g)$ * **Formation of Water:** $2H_2(g) + O_2(g) \rightarrow 2H_2O(l)$ * **Formation of Slaked Lime:** $CaO(s) + H_2O(l) \rightarrow Ca(OH)_2(aq) + \text{Heat}$ * *Quicklime (Calcium Oxide)* reacts vigorously with water to produce *Slaked Lime (Calcium Hydroxide)*. This is an **exothermic** reaction. * *Use of Slaked Lime:* Calcium hydroxide solution (limewater) is used for whitewashing walls. After 2-3 days, calcium hydroxide reacts slowly with carbon dioxide in the air to form a thin layer of calcium carbonate on the walls, giving them a shiny finish. $$ Ca(OH)_2(aq) + CO_2(g) \rightarrow CaCO_3(s) + H_2O(l) $$ #### 2. Decomposition Reaction A **decomposition reaction** is a reaction in which a single reactant breaks down into two or more simpler products. *General Form:* $AB \rightarrow A + B$ These reactions often require energy in the form of heat, light, or electricity to break the bonds of the reactant. ##### a. Thermal Decomposition (Decomposition by Heat) *Examples:* * **Decomposition of Ferrous Sulphate:** * When heated, green ferrous sulphate crystals ($FeSO_4 \cdot 7H_2O$) lose water and then decompose to form reddish-brown solid ferric oxide, sulphur dioxide, and sulphur trioxide. * $$ 2FeSO_4(s) \xrightarrow{\text{Heat}} Fe_2O_3(s) + SO_2(g) + SO_3(g) $$ * **Decomposition of Limestone:** * Calcium carbonate (limestone) decomposes on heating to form quicklime (calcium oxide) and carbon dioxide. This reaction is used in various industries, e.g., cement manufacturing. * $$ CaCO_3(s) \xrightarrow{\text{Heat}} CaO(s) + CO_2(g) $$ * **Decomposition of Lead Nitrate (Activity 1.5):** * When solid lead nitrate is heated, it decomposes to form yellow lead oxide, reddish-brown fumes of nitrogen dioxide, and oxygen gas. * $$ 2Pb(NO_3)_2(s) \xrightarrow{\text{Heat}} 2PbO(s) + 4NO_2(g) + O_2(g) $$ ##### b. Electrolytic Decomposition (Decomposition by Electricity) *Example:* * **Electrolysis of Water (Activity 1.7):** * When electric current is passed through acidified water, it decomposes into hydrogen gas and oxygen gas. Hydrogen gas is collected at the cathode, and oxygen gas at the anode. The volume of hydrogen gas collected is double that of oxygen gas. * $$ 2H_2O(l) \xrightarrow{\text{Electricity}} 2H_2(g) + O_2(g) $$ * *Diagram:* ##### c. Photolytic Decomposition (Decomposition by Light) *Examples:* * **Decomposition of Silver Chloride (Activity 1.8):** * Silver chloride turns greyish-white when exposed to sunlight due to its decomposition into silver metal and chlorine gas. * $$ 2AgCl(s) \xrightarrow{\text{Sunlight}} 2Ag(s) + Cl_2(g) $$ * **Decomposition of Silver Bromide:** * $$ 2AgBr(s) \xrightarrow{\text{Sunlight}} 2Ag(s) + Br_2(g) $$ * *Use:* These reactions are used in black and white photography. #### 3. Displacement Reaction A **displacement reaction** is a reaction in which a more reactive element displaces a less reactive element from its compound. *General Form:* $A + BC \rightarrow AC + B$ *Examples:* * **Iron Nail in Copper Sulphate Solution (Activity 1.9):** * When an iron nail is dipped in copper sulphate solution, the blue colour of copper sulphate solution fades, and a reddish-brown coating of copper metal is deposited on the iron nail. * $$ Fe(s) + CuSO_4(aq) \rightarrow FeSO_4(aq) + Cu(s) $$ * *Explanation:* Iron is more reactive than copper, so it displaces copper from copper sulphate solution. Iron sulphate solution is green. * **Zinc with Copper Sulphate:** * $$ Zn(s) + CuSO_4(aq) \rightarrow ZnSO_4(aq) + Cu(s) $$ * **Lead with Copper Chloride:** * $$ Pb(s) + CuCl_2(aq) \rightarrow PbCl_2(aq) + Cu(s) $$ * *Note:* Zinc and lead are more reactive elements than copper. They displace copper from its compounds. #### 4. Double Displacement Reaction A **double displacement reaction** is a reaction in which two different atoms or groups of atoms (ions) are exchanged between two compounds to form two new compounds. *General Form:* $AB + CD \rightarrow AD + CB$ *Types of Double Displacement Reactions:* ##### a. Precipitation Reactions Reactions that produce an insoluble solid (precipitate) are called **precipitation reactions**. *Example:* * **Reaction of Sodium Sulphate and Barium Chloride (Activity 1.10):** * When solutions of sodium sulphate and barium chloride are mixed, a white precipitate of barium sulphate is formed along with sodium chloride solution. * $$ Na_2SO_4(aq) + BaCl_2(aq) \rightarrow BaSO_4(s) \downarrow + 2NaCl(aq) $$ * *Ionic Equation:* $2Na^+(aq) + SO_4^{2-}(aq) + Ba^{2+}(aq) + 2Cl^-(aq) \rightarrow BaSO_4(s) + 2Na^+(aq) + 2Cl^-(aq)$ * *Net Ionic Equation:* $Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)$ ##### b. Neutralisation Reactions Reactions between an acid and a base to form salt and water are called **neutralisation reactions**. *Example:* * **Reaction of Hydrochloric Acid and Sodium Hydroxide:** * $$ HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l) $$ #### 5. Oxidation and Reduction Reactions (Redox Reactions) ##### Oxidation **Oxidation** is a process that involves: * Addition of oxygen to a substance. * Removal of hydrogen from a substance. * Loss of electrons. *Examples:* * $C + O_2 \rightarrow CO_2$ (Carbon is oxidised) * $2Cu + O_2 \xrightarrow{\text{Heat}} 2CuO$ (Copper is oxidised to copper oxide) * $H_2S + Cl_2 \rightarrow 2HCl + S$ (Hydrogen sulphide is oxidised to sulphur) ##### Reduction **Reduction** is a process that involves: * Removal of oxygen from a substance. * Addition of hydrogen to a substance. * Gain of electrons. *Examples:* * $CuO + H_2 \xrightarrow{\text{Heat}} Cu + H_2O$ (Copper oxide is reduced to copper) * $Cl_2 + H_2S \rightarrow 2HCl + S$ (Chlorine is reduced to hydrogen chloride) ##### Redox Reactions A **redox reaction** is a chemical reaction in which both oxidation and reduction occur simultaneously. *Example:* * $CuO(s) + H_2(g) \xrightarrow{\text{Heat}} Cu(s) + H_2O(l)$ * Here, $CuO$ is losing oxygen, so it is **reduced**. * $H_2$ is gaining oxygen, so it is **oxidised**. **Oxidising Agent:** The substance that oxidises another substance and itself gets reduced. (It provides oxygen or removes hydrogen). *In the above example:* $CuO$ is the oxidising agent. **Reducing Agent:** The substance that reduces another substance and itself gets oxidised. (It removes oxygen or provides hydrogen). *In the above example:* $H_2$ is the reducing agent. **Activity 1.11:** Oxidation of Copper * **Observation:** When a copper powder is heated in a china dish, its surface becomes coated with a black substance. * **Explanation:** This black substance is copper(II) oxide. It is formed due to the oxidation of copper by air (oxygen). $$ 2Cu(s) + O_2(g) \xrightarrow{\text{Heat}} 2CuO(s) $$ * **Reversing the reaction:** If hydrogen gas is passed over the heated black copper(II) oxide, the black coating turns brown again as copper is reformed. $$ CuO(s) + H_2(g) \xrightarrow{\text{Heat}} Cu(s) + H_2O(l) $$ * Here, $CuO$ is reduced to $Cu$ (by losing oxygen). * $H_2$ is oxidised to $H_2O$ (by gaining oxygen). ### Effects of Oxidation-Reduction Reactions in Everyday Life #### 1. Corrosion **Corrosion** is the process in which metals are eaten up gradually by the action of air, moisture, or a chemical (like an acid) on their surface. It is an oxidation reaction. *Examples:* * **Rusting of Iron:** When iron articles are exposed to moist air for a long time, they acquire a coating of a reddish-brown flaky substance called rust ($Fe_2O_3 \cdot xH_2O$). * $$ 4Fe(s) + 3O_2(g) + xH_2O(l) \rightarrow 2Fe_2O_3 \cdot xH_2O(s) $$ * **Black coating on Silver:** Silver articles become black after some time when exposed to air because they react with sulphur in the air to form silver sulphide ($Ag_2S$). * **Green coating on Copper:** Copper articles acquire a green coating when exposed to moist air for a long time. This is due to the formation of basic copper carbonate ($CuCO_3 \cdot Cu(OH)_2$). **Harmful Effects of Corrosion:** Corrosion causes damage to car bodies, bridges, iron railings, ships, and to all objects made of metals, especially iron. **Prevention of Corrosion:** * Painting * Oiling/Greasing * Galvanising (coating with zinc) * Electroplating * Alloying #### 2. Rancidity **Rancidity** is the oxidation of fats and oils present in food items, leading to a change in their smell and taste. * When fats and oils are exposed to air, they get oxidised, becoming rancid. **Prevention of Rancidity:** * **Adding Antioxidants:** Antioxidants are substances that prevent oxidation. * *Examples:* BHA (Butylated Hydroxyanisole) and BHT (Butylated Hydroxytoluene) are common antioxidants added to food containing fats and oils. * **Flushing with Nitrogen Gas:** Chips manufacturers usually flush bags of chips with nitrogen gas to prevent the chips from getting oxidised. Nitrogen is an unreactive gas. * **Refrigeration:** Keeping food in refrigerators slows down the oxidation process. * **Storing food in airtight containers:** This prevents oxygen from coming into contact with food. * **Storing food away from light:** Light can accelerate oxidation reactions. ### Extra Notes and Important Points * **Word Equation:** A simple way to represent a chemical reaction using names of reactants and products. * *Example:* Zinc + Sulphuric Acid $\rightarrow$ Zinc Sulphate + Hydrogen * **Skeletal Chemical Equation:** An unbalanced chemical equation using chemical formulae. * *Example:* $Zn + H_2SO_4 \rightarrow ZnSO_4 + H_2$ * **Exothermic Reactions:** Reactions that release energy, usually in the form of heat. * *Examples:* Burning of natural gas ($CH_4(g) + 2O_2(g) \rightarrow CO_2(g) + 2H_2O(l) + \text{Heat}$), Respiration ($C_6H_{12}O_6(aq) + 6O_2(g) \rightarrow 6CO_2(aq) + 6H_2O(l) + \text{Energy}$). * **Endothermic Reactions:** Reactions that absorb energy, usually in the form of heat or light. * *Examples:* Melting of ice, Photosynthesis ($6CO_2(g) + 6H_2O(l) \xrightarrow{\text{Sunlight}} C_6H_{12}O_6(aq) + 6O_2(g)$), Decomposition reactions. * **Photosynthesis:** A process where green plants convert light energy into chemical energy to synthesize food from carbon dioxide and water. * $$ 6CO_2(g) + 6H_2O(l) \xrightarrow{\text{Sunlight}} C_6H_{12}O_6(aq) + 6O_2(g) $$ * **Respiration:** The process by which living organisms release energy from food. It is an oxidation reaction and an exothermic process. * $$ C_6H_{12}O_6(aq) + 6O_2(g) \rightarrow 6CO_2(aq) + 6H_2O(l) + \text{Energy} $$ ### Mind Map: Chemical Reactions and Equations *Self-correction: As an AI, I cannot generate an actual image. The above is a placeholder. A typical mind map would have "Chemical Reactions and Equations" at the center, branching into "Characteristics," "Chemical Equations (Balanced, Unbalanced)," "Types of Reactions (Combination, Decomposition, Displacement, Double Displacement, Redox)," and "Effects of Redox (Corrosion, Rancidity)." Each of these branches would further extend to examples and sub-points.* ### Revision Table: Key Concepts | Concept | Definition/Explanation | Example(s) | | :----------------------- | :------------------------------------------------------------------------------------------- | :-------------------------------------------------------------------------------------------------- | | **Chemical Reaction** | Process involving rearrangement of molecular/ionic structure. | Burning of magnesium, rusting of iron. | | **Chemical Equation** | Symbolic representation of a chemical reaction. | $2Mg(s) + O_2(g) \rightarrow 2MgO(s)$ | | **Balanced Equation** | Same number of atoms of each element on both sides (Law of Conservation of Mass). | $3Fe(s) + 4H_2O(g) \rightarrow Fe_3O_4(s) + 4H_2(g)$ | | **Combination Reaction** | Two or more reactants combine to form a single product. | $CaO(s) + H_2O(l) \rightarrow Ca(OH)_2(aq)$ | | **Decomposition Reaction** | Single reactant breaks into two or more simpler products (requires energy). | $2FeSO_4(s) \xrightarrow{\text{Heat}} Fe_2O_3(s) + SO_2(g) + SO_3(g)$ | | **Thermal Decomposition**| Decomposition by heat. | $CaCO_3(s) \xrightarrow{\text{Heat}} CaO(s) + CO_2(g)$ | | **Electrolytic Decomposition** | Decomposition by electricity. | $2H_2O(l) \xrightarrow{\text{Electricity}} 2H_2(g) + O_2(g)$ | | **Photolytic Decomposition** | Decomposition by light. | $2AgCl(s) \xrightarrow{\text{Sunlight}} 2Ag(s) + Cl_2(g)$ | | **Displacement Reaction**| More reactive element displaces less reactive element from its compound. | $Fe(s) + CuSO_4(aq) \rightarrow FeSO_4(aq) + Cu(s)$ | | **Double Displacement Reaction** | Exchange of ions between two compounds to form two new compounds. | $Na_2SO_4(aq) + BaCl_2(aq) \rightarrow BaSO_4(s) + 2NaCl(aq)$ | | **Precipitation Reaction**| Double displacement reaction forming an insoluble product (precipitate). | $Pb(NO_3)_2(aq) + 2KI(aq) \rightarrow PbI_2(s) \downarrow + 2KNO_3(aq)$ | | **Neutralisation Reaction** | Acid reacts with base to form salt and water. | $HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l)$ | | **Oxidation** | Addition of oxygen, removal of hydrogen, or loss of electrons. | $2Cu + O_2 \rightarrow 2CuO$ | | **Reduction** | Removal of oxygen, addition of hydrogen, or gain of electrons. | $CuO + H_2 \rightarrow Cu + H_2O$ | | **Redox Reaction** | Oxidation and reduction occur simultaneously. | $CuO(s) + H_2(g) \rightarrow Cu(s) + H_2O(l)$ | | **Corrosion** | Gradual eating up of metals by air, moisture, or chemicals (oxidation). | Rusting of iron ($Fe_2O_3 \cdot xH_2O$) | | **Rancidity** | Oxidation of fats and oils in food, leading to bad smell/taste. | Chips becoming stale if not packed in nitrogen. | | **Exothermic Reaction** | Releases energy (heat). | Respiration, burning of natural gas. | | **Endothermic Reaction** | Absorbs energy (heat/light). | Photosynthesis, decomposition of $CaCO_3$. | ### Questions and Answers **Q1:** Why should a magnesium ribbon be cleaned before burning in air? **A1:** Magnesium ribbon should be cleaned with sandpaper before burning to remove the protective layer of basic magnesium carbonate ($MgCO_3 \cdot Mg(OH)_2$) or magnesium oxide ($MgO$) formed on its surface. This layer prevents the magnesium from coming into contact with oxygen and readily burning. Cleaning ensures pure magnesium is exposed to air for reaction. **Q2:** Write a balanced chemical equation with state symbols for the following reactions: a) Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and solution of sodium chloride. b) Sodium hydroxide solution (in water) reacts with hydrochloric acid solution (in water) to produce sodium chloride solution and water. **A2:** a) $BaCl_2(aq) + Na_2SO_4(aq) \rightarrow BaSO_4(s) \downarrow + 2NaCl(aq)$ b) $NaOH(aq) + HCl(aq) \rightarrow NaCl(aq) + H_2O(l)$ **Q3:** What is a balanced chemical equation? Why is it important to balance chemical equations? **A3:** A **balanced chemical equation** is a chemical equation in which the number of atoms of each element is the same on both the reactant side and the product side. It is important to balance chemical equations to satisfy the **Law of Conservation of Mass**. This law states that mass can neither be created nor destroyed in a chemical reaction. Therefore, the total mass of the elements present in the products of a chemical reaction must be equal to the total mass of the elements present in the reactants. Balancing ensures that the number of atoms of each element remains conserved throughout the reaction. **Q4:** Explain the following in terms of gain or loss of oxygen with two examples each: a) Oxidation b) Reduction **A4:** a) **Oxidation:** It is the process of addition of oxygen to a substance. * *Example 1:* $2Cu(s) + O_2(g) \xrightarrow{\text{Heat}} 2CuO(s)$ (Copper is oxidised to copper oxide) * *Example 2:* $C(s) + O_2(g) \rightarrow CO_2(g)$ (Carbon is oxidised to carbon dioxide) b) **Reduction:** It is the process of removal of oxygen from a substance. * *Example 1:* $CuO(s) + H_2(g) \xrightarrow{\text{Heat}} Cu(s) + H_2O(l)$ (Copper oxide is reduced to copper) * *Example 2:* $ZnO(s) + C(s) \xrightarrow{\text{Heat}} Zn(s) + CO(g)$ (Zinc oxide is reduced to zinc) **Q5:** What do you mean by an exothermic and an endothermic reaction? Give examples. **A5:** * **Exothermic Reactions:** Chemical reactions that release energy, primarily in the form of heat, are called exothermic reactions. The temperature of the surroundings increases during these reactions. * *Example 1:* Burning of natural gas: $CH_4(g) + 2O_2(g) \rightarrow CO_2(g) + 2H_2O(l) + \text{Heat}$ * *Example 2:* Respiration: $C_6H_{12}O_6(aq) + 6O_2(g) \rightarrow 6CO_2(aq) + 6H_2O(l) + \text{Energy}$ * **Endothermic Reactions:** Chemical reactions that absorb energy, primarily in the form of heat or light, from the surroundings are called endothermic reactions. The temperature of the surroundings decreases during these reactions. * *Example 1:* Decomposition of calcium carbonate: $CaCO_3(s) \xrightarrow{\text{Heat}} CaO(s) + CO_2(g)$ * *Example 2:* Photosynthesis: $6CO_2(g) + 6H_2O(l) \xrightarrow{\text{Sunlight}} C_6H_{12}O_6(aq) + 6O_2(g)$ **Q6:** Why are chips packets flushed with nitrogen gas? **A6:** Chips packets are flushed with nitrogen gas to prevent the chips from becoming rancid. Chips contain fats and oils, which can get oxidised when exposed to oxygen in the air, leading to a change in their smell and taste (rancidity). Nitrogen is an unreactive gas and acts as an antioxidant, displacing oxygen and thus preventing the oxidation of fats and oils in the chips, keeping them fresh for a longer time. **Q7:** Describe the difference between a displacement reaction and a double displacement reaction. Write equations for each. **A7:** * **Displacement Reaction:** In a displacement reaction, a more reactive element displaces a less reactive element from its compound. There is a "single" exchange of elements. * *General Form:* $A + BC \rightarrow AC + B$ * *Example:* $Zn(s) + CuSO_4(aq) \rightarrow ZnSO_4(aq) + Cu(s)$ * **Double Displacement Reaction:** In a double displacement reaction, there is an exchange of ions (atoms or groups of atoms) between two compounds to form two new compounds. * *General Form:* $AB + CD \rightarrow AD + CB$ * *Example:* $Na_2SO_4(aq) + BaCl_2(aq) \rightarrow BaSO_4(s) \downarrow + 2NaCl(aq)$ **Q8:** What happens when dilute hydrochloric acid is added to iron fillings? Mark the correct answer. (a) Hydrogen gas and iron chloride are produced. (b) Chlorine gas and iron hydroxide are produced. (c) No reaction takes place. (d) Iron salt and water are produced. **A8:** (a) Hydrogen gas and iron chloride are produced. * Equation: $Fe(s) + 2HCl(aq) \rightarrow FeCl_2(aq) + H_2(g) \uparrow$ **Q9:** What is meant by precipitation reaction? Explain with an example. **A9:** A **precipitation reaction** is a chemical reaction that produces an insoluble solid, called a precipitate, when two soluble solutions are mixed. The precipitate separates out from the solution. * *Example:* When a solution of potassium iodide is added to a solution of lead nitrate, a yellow precipitate of lead iodide is formed. $$ Pb(NO_3)_2(aq) + 2KI(aq) \rightarrow PbI_2(s) \downarrow + 2KNO_3(aq) $$ Here, $PbI_2$ is the yellow precipitate.