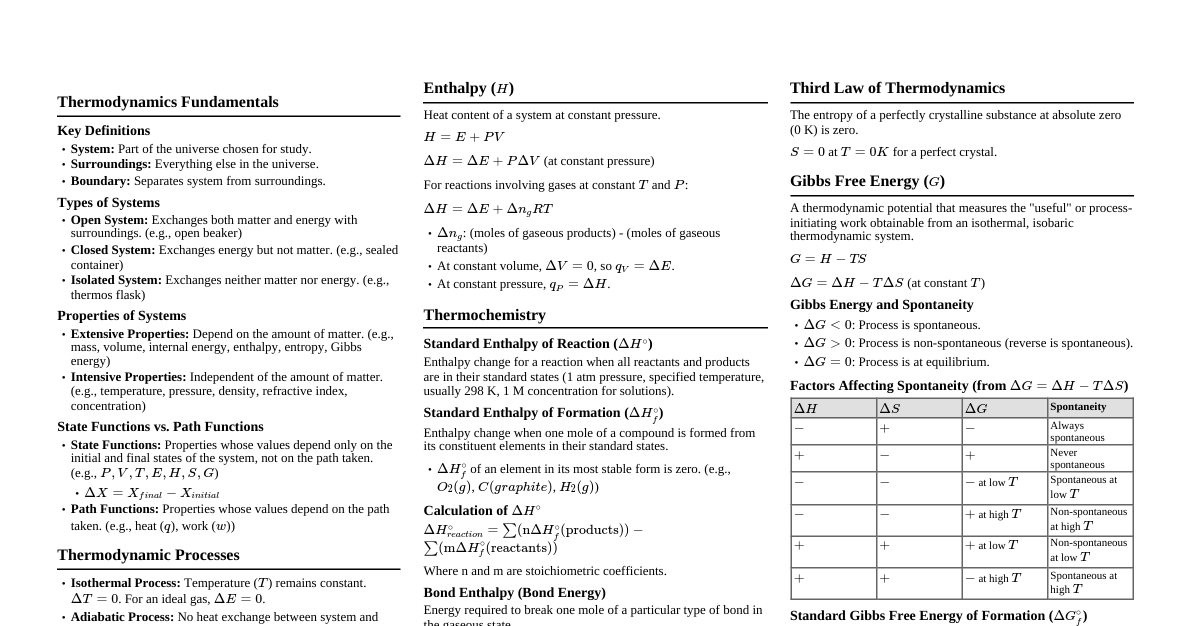
### Thermodynamic Systems & Processes - **System:** Part of the universe chosen for study. - **Open:** Exchanges both energy and matter with surroundings. - **Closed:** Exchanges energy but not matter. - **Isolated:** Exchanges neither energy nor matter. - **Surroundings:** Everything outside the system. - **Boundary:** Separates system from surroundings. - **Thermodynamic Variables:** Pressure (P), Volume (V), Temperature (T), Internal Energy (U), Entropy (S). - **Thermodynamic Process:** Change in state of a system. - **Isothermal:** T = constant ($\Delta T = 0$). For ideal gas, $PV = \text{constant}$. $W = nRT \ln(V_f/V_i) = 2.303 nRT \log(V_f/V_i)$. - **Adiabatic:** No heat exchange (Q = 0). $PV^\gamma = \text{constant}$, $T V^{\gamma-1} = \text{constant}$, $T^\gamma P^{1-\gamma} = \text{constant}$. $W = \frac{nR(T_i - T_f)}{\gamma - 1}$. - **Isobaric:** P = constant ($\Delta P = 0$). $W = P(V_f - V_i)$. - **Isochoric:** V = constant ($\Delta V = 0$). $W = 0$. - **Cyclic Process:** Initial and final states are the same. $\Delta U = 0$, $Q = W$. - **Reversible Process:** Can be reversed without leaving any change in the surroundings. - **Irreversible Process:** Cannot be reversed without leaving changes in the surroundings (most natural processes are irreversible). ### First Law of Thermodynamics (FLOT) - **Statement:** Energy cannot be created or destroyed, only transformed. - **Equation:** $\Delta Q = \Delta U + \Delta W$ - $\Delta Q$: Heat supplied to the system (positive if absorbed, negative if released). - $\Delta U$: Change in internal energy (positive if increases, negative if decreases). - $\Delta W$: Work done by the system (positive if done by system, negative if done on system). - **Internal Energy (U):** Depends only on temperature for an ideal gas. $\Delta U = n C_v \Delta T$. - **Work Done (W):** Area under P-V curve. $W = \int P dV$. - Expansion: $W > 0$. - Compression: $W 0$. The slope of P-V curve is steepest for adiabatic, then isothermal, then isobaric. - **Solution:** $W_1 > W_2 > W_3$. (Draw P-V diagram to visualize areas). - **NEET 2016 (Phase II):** One mole of an ideal monatomic gas undergoes a process where $P=aV^2$. The temperature of the gas changes from $T_0$ to $2T_0$. The heat supplied to the gas is... - **Concept:** Use FLOT: $Q = \Delta U + W$. $\Delta U = n C_V \Delta T$. Work $W = \int P dV = \int aV^2 dV$. - **Solution:** $P_1 = aV_1^2$, $P_2 = aV_2^2$. $PV = nRT \implies aV^3 = nRT$. $T_0 = aV_1^3/(nR)$, $2T_0 = aV_2^3/(nR) \implies V_2^3 = 2V_1^3 \implies V_2 = V_1 2^{1/3}$. $W = \int_{V_1}^{V_2} aV^2 dV = a[\frac{V^3}{3}]_{V_1}^{V_2} = \frac{a}{3}(V_2^3 - V_1^3) = \frac{a}{3}(2V_1^3 - V_1^3) = \frac{aV_1^3}{3}$. Substitute $aV_1^3 = nRT_0$: $W = \frac{nRT_0}{3}$. For monatomic gas, $C_V = \frac{3}{2}R$. $\Delta U = n C_V \Delta T = n (\frac{3}{2}R)(2T_0 - T_0) = \frac{3}{2}nRT_0$. $Q = \Delta U + W = \frac{3}{2}nRT_0 + \frac{nRT_0}{3} = (\frac{9+2}{6})nRT_0 = \frac{11}{6}nRT_0$. - **NEET 2015:** An ideal gas is compressed isothermally. What is the change in internal energy and heat supplied? - **Concept:** Isothermal process means T=constant. For ideal gas, $\Delta U = 0$. - **Solution:** $\Delta U = 0$. Since work is done *on* the gas (compression), $W$ is negative. From $Q = \Delta U + W$, $Q=W$. Since $W$ is negative, $Q$ is negative, meaning heat is released. - **NEET 2014:** A thermodynamic system performs work of $100J$ when $200J$ of heat is supplied to it. The change in internal energy is... - **Concept:** Direct application of FLOT: $\Delta U = Q - W$. - **Solution:** $\Delta U = 200J - 100J = 100J$. - **NEET 2013:** An ideal gas undergoes an adiabatic expansion. What is the relation between pressure and volume? - **Concept:** Definition of adiabatic process for ideal gas. - **Solution:** $PV^\gamma = \text{constant}$. - **NEET 2012:** If the ratio of specific heats of a gas at constant pressure to constant volume is $\gamma$, the change in internal energy of a mass of gas when the volume changes from V to 2V at constant pressure P is... - **Concept:** $\Delta U = n C_V \Delta T$. For constant pressure, $P V = n R T \implies P \Delta V = n R \Delta T$. - **Solution:** $P(2V - V) = n R \Delta T \implies PV = n R \Delta T \implies \Delta T = PV/(nR)$. $\Delta U = n C_V \Delta T = n C_V \frac{PV}{nR} = \frac{C_V}{R} PV$. Since $C_P - C_V = R \implies R = C_V(\gamma - 1)$, then $\frac{C_V}{R} = \frac{1}{\gamma - 1}$. So, $\Delta U = \frac{PV}{\gamma - 1}$. ### Specific Heat Capacities of Gases - **Molar Specific Heat ($C$):** Heat required to raise temperature of 1 mole of substance by $1^\circ C$. - $C_P$: Molar specific heat at constant pressure. - $C_V$: Molar specific heat at constant volume. - **Relation between $C_P$ and $C_V$ (Mayer's Formula):** $C_P - C_V = R$. - **Ratio of Specific Heats ($\gamma$):** $\gamma = C_P / C_V$. - Monatomic gas (He, Ne, Ar): $C_V = \frac{3}{2}R$, $C_P = \frac{5}{2}R$, $\gamma = \frac{5}{3} \approx 1.67$. - Diatomic gas (O$_2$, N$_2$, H$_2$): $C_V = \frac{5}{2}R$, $C_P = \frac{7}{2}R$, $\gamma = \frac{7}{5} = 1.4$. (At room temp, neglecting vibrational modes). - Polyatomic gas (CO$_2$, NH$_3$): $C_V = 3R$, $C_P = 4R$, $\gamma = \frac{4}{3} \approx 1.33$. (General case). #### **NEET Focus:** - **★ Important:** Applying Mayer's formula and values of $\gamma$ for different gases. - **★ Important:** Calculating $\Delta U = n C_V \Delta T$ and $Q = n C \Delta T$. ### Heat Engines & Refrigerators - **Heat Engine:** Converts heat energy into mechanical work. - **Efficiency ($\eta$):** $\eta = \frac{\text{Work Done}}{\text{Heat Input}} = \frac{W}{Q_H} = \frac{Q_H - Q_C}{Q_H} = 1 - \frac{Q_C}{Q_H}$. - $Q_H$: Heat absorbed from hot reservoir. - $Q_C$: Heat rejected to cold reservoir. - **Carnot Engine (Ideal Engine):** Most efficient engine. - **Carnot Efficiency:** $\eta_{Carnot} = 1 - \frac{T_C}{T_H}$. (Temperatures must be in Kelvin). - **Carnot Cycle:** Two isothermal and two adiabatic processes. - **Refrigerator/Heat Pump:** Moves heat from cold to hot reservoir (work must be done on the system). - **Coefficient of Performance (COP):** - **Refrigerator:** $COP_R = \frac{\text{Heat Extracted from Cold Reservoir}}{\text{Work Done}} = \frac{Q_C}{W} = \frac{Q_C}{Q_H - Q_C}$. - **Heat Pump:** $COP_{HP} = \frac{\text{Heat Delivered to Hot Reservoir}}{\text{Work Done}} = \frac{Q_H}{W} = \frac{Q_H}{Q_H - Q_C}$. - **Relation:** $COP_{HP} = COP_R + 1$. - **Carnot COP:** - $COP_{R,Carnot} = \frac{T_C}{T_H - T_C}$. - $COP_{HP,Carnot} = \frac{T_H}{T_H - T_C}$. #### **NEET Focus:** - **★ Important:** Calculations of efficiency and COP using given heat values or temperatures. - Understanding the difference between heat engine and refrigerator. ### Second Law of Thermodynamics - **Clausius Statement:** Heat cannot flow spontaneously from a colder body to a hotter body without some external work being done. - **Kelvin-Planck Statement:** It is impossible to construct a heat engine that operates in a cycle and produces no other effect than the extraction of heat from a reservoir and the performance of an equivalent amount of work. (No engine can have 100% efficiency). - **Entropy (S):** Measure of disorder or randomness of a system. - **Change in Entropy:** $\Delta S = \frac{\Delta Q_{rev}}{T}$. For a reversible process. - **Total Entropy for isolated system in a reversible process:** $\Delta S_{total} = \Delta S_{system} + \Delta S_{surroundings} = 0$. - **Total Entropy for isolated system in an irreversible process:** $\Delta S_{total} = \Delta S_{system} + \Delta S_{surroundings} > 0$. - **Principle of Increase of Entropy:** The entropy of an isolated system never decreases; it either increases (for irreversible processes) or remains constant (for reversible processes). #### **NEET Focus:** - Conceptual understanding of statements and entropy. - Qualitative questions on entropy change for different processes (e.g., melting ice, gas expansion). ### Zeroth Law of Thermodynamics - **Statement:** If two systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. - **Importance:** Defines temperature as a fundamental property. ### Important Formulas and Relations - **Work done for various processes (ideal gas):** - Isothermal: $W = nRT \ln(V_f/V_i)$ - Adiabatic: $W = \frac{P_iV_i - P_fV_f}{\gamma - 1} = \frac{nR(T_i - T_f)}{\gamma - 1}$ - Isobaric: $W = P(V_f - V_i)$ - Isochoric: $W = 0$ - **Internal Energy Change:** $\Delta U = n C_V \Delta T = \frac{n R \Delta T}{\gamma - 1}$. - **Degrees of Freedom ($f$):** - Monatomic: $f=3$ (translational) - Diatomic: $f=5$ (3 translational + 2 rotational) - Polyatomic: $f=6$ (3 translational + 3 rotational) - **Equipartition of Energy:** Energy per degree of freedom = $\frac{1}{2}kT$ (per molecule) or $\frac{1}{2}RT$ (per mole). - **Relation between $C_V$ and $f$:** $C_V = \frac{f}{2}R$. - **Relation between $C_P$ and $f$:** $C_P = (\frac{f}{2} + 1)R$. - **Relation between $\gamma$ and $f$:** $\gamma = 1 + \frac{2}{f}$. #### **NEET Focus:** - **★ Important:** Direct application of these formulas in problem-solving. - Understanding the relationship between degrees of freedom and specific heats.