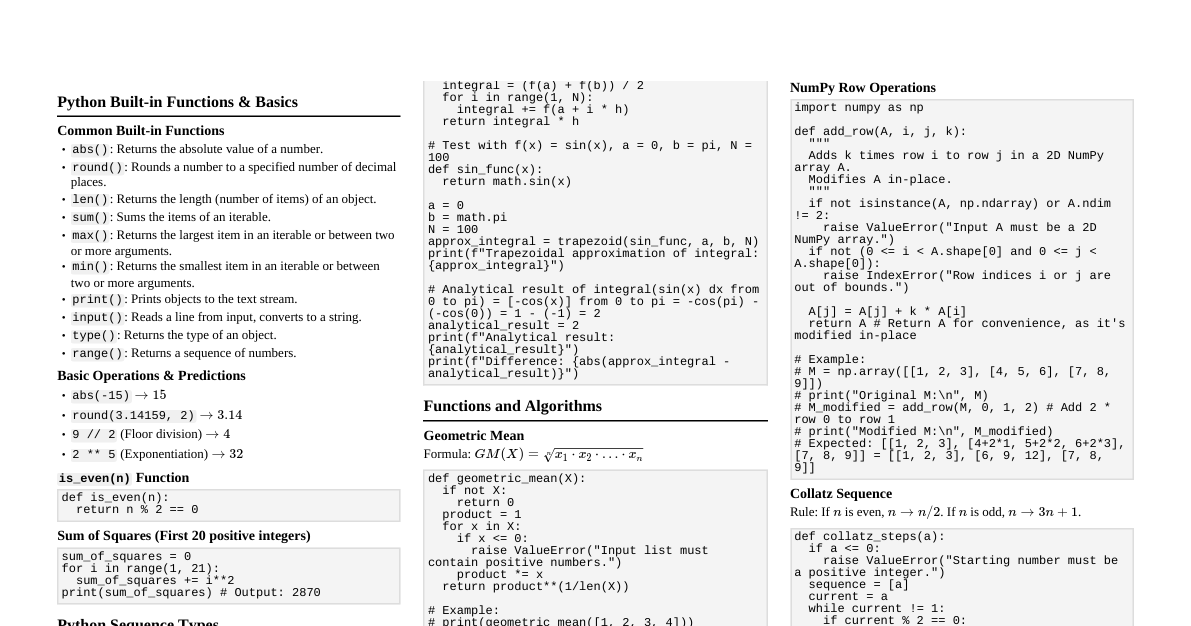
### Mathematics Fundamentals #### Algebra * **Quadratic Formula:** For $ax^2 + bx + c = 0$, $x = \frac{-b \pm \sqrt{b^2 - 4ac}}{2a}$ * **Logarithm Properties:** * $\log_b(MN) = \log_b(M) + \log_b(N)$ * $\log_b(M/N) = \log_b(M) - \log_b(N)$ * $\log_b(M^k) = k \log_b(M)$ * $\log_b(b) = 1$, $\log_b(1) = 0$ * Change of Base: $\log_b(x) = \frac{\log_c(x)}{\log_c(b)}$ #### Trigonometry * **SOH CAH TOA:** * $\sin(\theta) = \text{Opposite}/\text{Hypotenuse}$ * $\cos(\theta) = \text{Adjacent}/\text{Hypotenuse}$ * $\tan(\theta) = \text{Opposite}/\text{Adjacent}$ * **Pythagorean Identity:** $\sin^2(\theta) + \cos^2(\theta) = 1$ * **Double Angle Formulas:** * $\sin(2\theta) = 2\sin(\theta)\cos(\theta)$ * $\cos(2\theta) = \cos^2(\theta) - \sin^2(\theta) = 2\cos^2(\theta) - 1 = 1 - 2\sin^2(\theta)$ #### Calculus - Differentiation * **Power Rule:** $\frac{d}{dx}(x^n) = nx^{n-1}$ * **Product Rule:** $\frac{d}{dx}(uv) = u'v + uv'$ * **Quotient Rule:** $\frac{d}{dx}\left(\frac{u}{v}\right) = \frac{u'v - uv'}{v^2}$ * **Chain Rule:** $\frac{d}{dx}(f(g(x))) = f'(g(x))g'(x)$ * **Common Derivatives:** * $\frac{d}{dx}(\sin x) = \cos x$ * $\frac{d}{dx}(\cos x) = -\sin x$ * $\frac{d}{dx}(e^x) = e^x$ * $\frac{d}{dx}(\ln x) = \frac{1}{x}$ #### Calculus - Integration * **Power Rule:** $\int x^n dx = \frac{x^{n+1}}{n+1} + C$ (for $n \ne -1$) * **$\int \frac{1}{x} dx = \ln|x| + C$** * **$\int e^x dx = e^x + C$** * **$\int \sin x dx = -\cos x + C$** * **$\int \cos x dx = \sin x + C$** * **Integration by Parts:** $\int u dv = uv - \int v du$ ### Physics - Mechanics #### Kinematics (Constant Acceleration) * $v = v_0 + at$ * $x = x_0 + v_0t + \frac{1}{2}at^2$ * $v^2 = v_0^2 + 2a(x - x_0)$ * $x = x_0 + \frac{1}{2}(v_0 + v)t$ #### Newton's Laws 1. **Inertia:** An object at rest stays at rest, and an object in motion stays in motion with the same speed and in the same direction unless acted upon by an unbalanced force. 2. **$F = ma$**: The acceleration of an object is directly proportional to the net force acting on it and inversely proportional to its mass. 3. **Action-Reaction:** For every action, there is an equal and opposite reaction. #### Work, Energy, Power * **Work:** $W = Fd\cos\theta$ * **Kinetic Energy:** $KE = \frac{1}{2}mv^2$ * **Gravitational Potential Energy:** $PE_g = mgh$ * **Elastic Potential Energy:** $PE_s = \frac{1}{2}kx^2$ * **Work-Energy Theorem:** $W_{net} = \Delta KE$ * **Conservation of Energy:** $KE_i + PE_i = KE_f + PE_f$ (for conservative forces) * **Power:** $P = \frac{W}{t} = Fv\cos\theta$ #### Momentum & Impulse * **Momentum:** $p = mv$ * **Impulse:** $J = F\Delta t = \Delta p$ * **Conservation of Momentum:** $m_1v_{1i} + m_2v_{2i} = m_1v_{1f} + m_2v_{2f}$ (in isolated systems) ### Chemistry - Fundamentals #### Stoichiometry * **Molar Mass:** Sum of atomic masses of all atoms in a molecule. * **Moles:** $n = \frac{\text{mass (g)}}{\text{molar mass (g/mol)}}$ * **Avogadro's Number:** $6.022 \times 10^{23}$ particles/mol * **Molarity:** $M = \frac{\text{moles of solute}}{\text{liters of solution}}$ #### Gas Laws * **Boyle's Law:** $P_1V_1 = P_2V_2$ (constant T, n) * **Charles's Law:** $\frac{V_1}{T_1} = \frac{V_2}{T_2}$ (constant P, n) * **Gay-Lussac's Law:** $\frac{P_1}{T_1} = \frac{P_2}{T_2}$ (constant V, n) * **Combined Gas Law:** $\frac{P_1V_1}{T_1} = \frac{P_2V_2}{T_2}$ * **Ideal Gas Law:** $PV = nRT$ (where $R = 0.0821 \frac{\text{L}\cdot\text{atm}}{\text{mol}\cdot\text{K}}$ or $8.314 \frac{\text{J}}{\text{mol}\cdot\text{K}}$) #### Acids and Bases * **Arrhenius:** Acid produces $H^+$, Base produces $OH^-$ * **Brønsted-Lowry:** Acid is $H^+$ donor, Base is $H^+$ acceptor * **pH Scale:** $pH = -\log[H^+]$ * **pOH Scale:** $pOH = -\log[OH^-]$ * **Relationship:** $pH + pOH = 14$ (at 25°C) * **Equilibrium Constant for Water:** $K_w = [H^+][OH^-] = 1.0 \times 10^{-14}$ (at 25°C) * **Acid Dissociation Constant:** $K_a = \frac{[H^+][A^-]}{[HA]}$ * **Base Dissociation Constant:** $K_b = \frac{[BH^+][OH^-]}{[B]}$ * **Relationship:** $K_a \cdot K_b = K_w$ (for conjugate acid-base pairs) ### Biology - Cell Structure & Function #### Prokaryotic vs. Eukaryotic Cells | Feature | Prokaryotic (Bacteria, Archaea) | Eukaryotic (Animals, Plants, Fungi, Protists) | |-------------------|---------------------------------|-----------------------------------------------| | **Size** | Small (1-10 µm) | Large (10-100 µm) | | **Nucleus** | No true nucleus (nucleoid region) | True nucleus (membrane-bound) | | **Organelles** | No membrane-bound organelles | Membrane-bound organelles (mitochondria, ER, Golgi) | | **DNA** | Circular, in cytoplasm | Linear, in nucleus (chromosomes) | | **Ribosomes** | Present (smaller) | Present (larger) | | **Cell Wall** | Present (peptidoglycan) | Present (plants, fungi), absent (animals) | #### Key Eukaryotic Organelles * **Nucleus:** Contains genetic material (DNA), controls cell activities. * **Mitochondria:** "Powerhouse of the cell", site of cellular respiration (ATP production). * **Endoplasmic Reticulum (ER):** * **Rough ER:** Studded with ribosomes, synthesizes and modifies proteins for secretion or insertion into membranes. * **Smooth ER:** Synthesizes lipids, detoxifies drugs/poisons, stores calcium ions. * **Golgi Apparatus:** Modifies, sorts, and packages proteins and lipids for secretion or delivery to other organelles. * **Lysosomes:** Contain hydrolytic enzymes to digest waste materials and cellular debris. * **Vacuoles:** (Large in plants) Storage, waste removal, maintain turgor pressure. * **Chloroplasts:** (Plants, algae) Site of photosynthesis. * **Cell Membrane:** Phospholipid bilayer, selectively permeable, regulates passage of substances. #### Cell Transport * **Passive Transport:** No energy required, movement down concentration gradient. * **Diffusion:** Movement of molecules from high to low concentration. * **Facilitated Diffusion:** Diffusion aided by transport proteins. * **Osmosis:** Diffusion of water across a selectively permeable membrane. * **Active Transport:** Requires energy (ATP), movement against concentration gradient. * **Pumps:** e.g., Sodium-Potassium pump. * **Bulk Transport:** * **Endocytosis:** Cell takes in substances (phagocytosis, pinocytosis, receptor-mediated). * **Exocytosis:** Cell expels substances. ### Computer Science - Basics #### Data Structures * **Arrays:** * Fixed-size collection of elements of the same type. * Access by index (O(1)). * Insertion/Deletion can be O(N). * **Linked Lists:** * Dynamic collection of nodes, each pointing to the next. * Insertion/Deletion O(1) (if position known). * Access O(N). * **Stacks (LIFO):** * `push()`: Add element to top. * `pop()`: Remove element from top. * **Queues (FIFO):** * `enqueue()`: Add element to rear. * `dequeue()`: Remove element from front. * **Trees:** * Hierarchical structure, root node, child nodes. * **Binary Search Trees (BST):** Left child ### Engineering - Thermodynamics #### Basic Concepts * **System:** A quantity of matter or a region in space chosen for study. * **Surroundings:** Everything external to the system. * **Boundary:** The real or imaginary surface separating the system from its surroundings. * **Closed System:** Mass cannot cross the boundary, but energy can. * **Open System (Control Volume):** Both mass and energy can cross the boundary. * **Isolated System:** Neither mass nor energy can cross the boundary. #### Laws of Thermodynamics 1. **Zeroth Law:** If two thermodynamic systems are each in thermal equilibrium with a third, then they are in thermal equilibrium with each other. (Defines temperature) 2. **First Law (Conservation of Energy):** * **Closed System:** $\Delta U = Q - W$ (Change in internal energy = Heat added - Work done by system) * **Open System (Steady Flow):** $\dot{Q} - \dot{W} = \sum_{out} \dot{m}(h + \frac{V^2}{2} + gz) - \sum_{in} \dot{m}(h + \frac{V^2}{2} + gz)$ * Where $h = u + Pv$ (enthalpy), $\dot{m}$ is mass flow rate. 3. **Second Law:** * **Kelvin-Planck Statement:** It is impossible for any device that operates on a cycle to receive heat from a single reservoir and produce a net amount of work. (No 100% efficient heat engine) * **Clausius Statement:** It is impossible to construct a device that operates in a cycle and produces no effect other than the transfer of heat from a lower-temperature body to a higher-temperature body. (Heat doesn't spontaneously flow cold to hot) * **Entropy:** $\Delta S \ge \int \frac{\delta Q}{T}$. For an isolated system, $\Delta S_{isolated} \ge 0$. (Entropy of isolated system never decreases) 4. **Third Law:** The entropy of a perfect crystal at absolute zero (0 K) is exactly zero. #### Thermodynamic Properties * **Temperature (T):** A measure of the average kinetic energy of the particles. * **Pressure (P):** Force per unit area. * **Volume (V):** Space occupied by the system. * **Internal Energy (U):** Energy associated with the microscopic components of a system. * **Enthalpy (H):** $H = U + PV$. Useful for constant pressure processes. * **Entropy (S):** A measure of the disorder or randomness of a system. * **Gibbs Free Energy (G):** $G = H - TS$. Predicts spontaneity of processes at constant T and P. * **Helmholtz Free Energy (A):** $A = U - TS$. Predicts spontaneity of processes at constant T and V.