Contraceptives: OCP & IUD
Cheatsheet Content
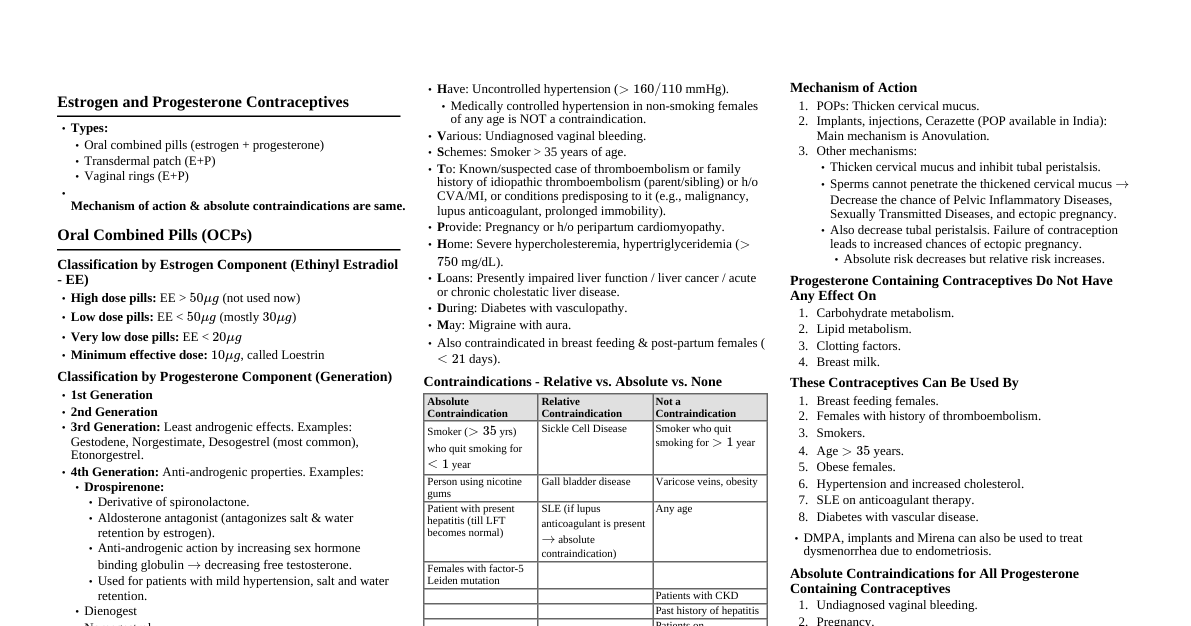
### Introduction to Contraception Contraception refers to methods or devices used to prevent pregnancy. Effective contraception is a cornerstone of reproductive health, family planning, and women's empowerment. This cheatsheet focuses on Oral Contraceptive Pills (OCPs) and Intrauterine Contraceptive Devices (IUCDs), two of the most widely used and effective reversible contraceptive methods. #### Importance of Contraception - Prevents unintended pregnancies - Reduces maternal and infant mortality - Allows for birth spacing and family planning - Manages certain gynecological conditions (e.g., dysmenorrhea, menorrhagia, endometriosis, PCOS) - Reduces risk of certain cancers (e.g., ovarian and endometrial with OCPs) ### Oral Contraceptive Pills (OCPs) Overview OCPs are hormonal medications taken orally to prevent pregnancy. They are highly effective when used correctly and consistently. #### Types of OCPs 1. **Combined Oral Contraceptives (COCs):** Contain both estrogen and progestin. * **Monophasic:** Each active pill contains the same amount of estrogen and progestin. * **Biphasic:** Mimic the natural menstrual cycle by altering the progestin-to-estrogen ratio once during the cycle. * **Triphasic:** Mimic the natural menstrual cycle by altering the progestin-to-estrogen ratio twice during the cycle. * **Quadriphasic:** Newer formulation with four different doses of estrogen and progestin. 2. **Progestin-Only Pills (POPs) / Mini-Pills:** Contain only progestin. * Primarily used when estrogen is contraindicated. #### Mechanisms of Action * **COCs:** * **Primary:** Suppress ovulation by inhibiting GnRH release, thereby preventing FSH and LH surges. * **Secondary:** Thicken cervical mucus (impeding sperm transport), thin the endometrial lining (inhibiting implantation). * **POPs:** * **Primary:** Thicken cervical mucus. * **Secondary:** Suppress ovulation (less consistently than COCs), thin the endometrial lining. ### OCPs: Pharmacology & Dosing Understanding the hormonal components and their metabolism is crucial for appropriate prescribing. #### Estrogen Components * **Ethinyl Estradiol (EE):** Most common synthetic estrogen. * **Estradiol Valerate:** Prodrug of estradiol, found in some newer COCs. * **Estetrol (E4):** A novel natural estrogen, found in a new COC. * **Dose:** Typically 20-35 µg EE in modern COCs. Lower doses (≤20 µg) may have fewer side effects but slightly higher breakthrough bleeding. #### Progestin Components Vary widely, influencing side effect profiles and non-contraceptive benefits. * **First Generation:** Norethindrone, ethynodiol diacetate. * **Second Generation:** Levonorgestrel, norgestrel. More androgenic. * **Third Generation:** Desogestrel, gestodene, norgestimate. Less androgenic, potentially higher VTE risk. * **Fourth Generation:** Drospirenone (anti-androgenic, anti-mineralocorticoid), dienogest (anti-androgenic), nestorone. #### Dosing Regimens * **Conventional (21/7 or 24/4):** 21-24 active pills followed by 4-7 placebo pills (or no pills) to induce withdrawal bleeding. * **Extended Cycle (84/7):** 84 active pills followed by 7 placebo pills, resulting in 4 periods per year. * **Continuous Use:** Active pills taken daily without a break, aiming for amenorrhea. Requires counseling on potential breakthrough bleeding. #### Pharmacokinetics * **Absorption:** Rapidly absorbed orally. * **Metabolism:** Primarily hepatic via cytochrome P450 enzymes (CYP3A4 for EE). Undergoes enterohepatic recirculation. * **Excretion:** Renal and fecal. * **Drug Interactions:** * **Enzyme Inducers:** Rifampicin, anticonvulsants (phenytoin, carbamazepine, barbiturates, topiramate), St. John's Wort. Decrease OCP efficacy. * **Enzyme Inhibitors:** Azole antifungals, protease inhibitors. May increase OCP levels. * **Antibiotics:** Most antibiotics (except rifampicin) do NOT significantly reduce OCP efficacy. ### OCPs: Non-Contraceptive Benefits & Risks #### Non-Contraceptive Benefits * **Menstrual Cycle Control:** * Reduced dysmenorrhea and menorrhagia. * Regular cycles, reduced premenstrual dysphoric disorder (PMDD). * **Reproductive Health:** * Reduced risk of ovarian and endometrial cancer. * Reduced risk of benign breast disease, ovarian cysts, ectopic pregnancy, PID. * **Androgen-Related Conditions (especially with anti-androgenic progestins like drospirenone/dienogest):** * Improved acne, hirsutism. * **Bone Health:** May preserve bone density in some premenopausal women. #### Risks & Side Effects * **Common Side Effects (usually transient, resolve in 3 months):** * Nausea, breast tenderness, breakthrough bleeding/spotting, headache, mood changes, weight changes (often perceived, not always actual). * **Serious Risks (Rare but significant):** * **Venous Thromboembolism (VTE):** 3-9 fold increased risk (absolute risk remains low, e.g., 6-12 per 10,000 woman-years vs. 1-5 for non-users). Risk influenced by estrogen dose, progestin type (3rd/4th gen may have slightly higher risk than 2nd gen), and individual risk factors. * **Arterial Thrombosis (MI, Stroke):** Increased risk in women with pre-existing risk factors (e.g., hypertension, smoking, migraine with aura, diabetes). * **Hypertension:** Small increase in blood pressure in some women. * **Gallbladder Disease:** Increased risk of gallstones. * **Liver Tumors:** Very rare, benign hepatic adenomas; extremely rare hepatocellular carcinoma. * **Cervical Cancer:** Small increased risk with long-term use (>5 years), likely due to confounding factors (e.g., HPV exposure). * **Breast Cancer:** Current evidence is conflicting; some studies show a small, transient increase in risk while on OCPs, but no increased risk after discontinuation. #### Contraindications (WHO Medical Eligibility Criteria - MEC) * **Category 4 (Absolute Contraindication):** * Current or history of DVT/PE * Major surgery with prolonged immobilization * Known thrombogenic mutations * Current or history of ischemic heart disease, stroke * Untreated hypertension (BP ≥ 160/100 mmHg) * Severe migraine with aura (at any age) * Current breast cancer (POPs can be used if benefits outweigh risks) * Severe decompensated cirrhosis, hepatocellular adenoma, malignant liver tumor * Diabetes with nephropathy/retinopathy/neuropathy or >20 years duration * Smoking ≥15 cigarettes/day and ≥35 years old * **Category 3 (Risks generally outweigh benefits):** * Controlled hypertension (BP 140-159/90-99 mmHg) * Migraine without aura (≥35 years old) * History of bariatric surgery * Gallbladder disease (symptomatic) * Past breast cancer (no recurrence for 5 years) * Postpartum ### OCPs: Prescribing & Management #### Initiation * **Quick Start:** Start immediately, use backup contraception for 7 days. Most common method. * **First Day of Menses:** Start on day 1-5 of cycle, no backup needed. * **Sunday Start:** Start on first Sunday after menses, use backup for 7 days. #### Missed Pills Management * **One missed pill ( 24 hours late):** * Take the most recent missed pill, discard earlier missed pills. * Continue remaining pills at usual time. * Use backup contraception for 7 days. * If pills missed in the last week of active pills (day 15-21 for 21-day pack, day 22-24 for 24-day pack), skip placebo pills and start new pack immediately. Consider emergency contraception if unprotected intercourse occurred in the last 5 days. #### POPs Specifics * **Strict Timing:** Must be taken at the same time every day (within a 3-hour window). * **Missed POP:** If >3 hours late, take as soon as remembered, use backup for 48 hours. Consider EC if unprotected intercourse occurred. * **Ideal for:** Breastfeeding women (after 6 weeks postpartum), women with estrogen contraindications. #### Counseling Points * Emphasize consistent daily use. * Discuss potential side effects and what to do. * Explain warning signs for serious complications (ACHES: Abdominal pain, Chest pain, Headaches, Eye problems, Severe leg pain). * Review drug interactions. * Advise on STI prevention (OCPs do not protect against STIs). ### Intrauterine Contraceptive Devices (IUCDs) Overview IUCDs are small, T-shaped devices inserted into the uterus to prevent pregnancy. They are highly effective, long-acting reversible contraceptives (LARCs). #### Types of IUCDs 1. **Copper IUCDs (Cu-IUCD):** Non-hormonal. * Examples: ParaGard (T380A), Nova-T, Multiload. * Duration of action: Up to 10-12 years. 2. **Hormonal IUCDs (Levonorgestrel-Releasing Intrauterine System - LNG-IUS):** * Examples: Mirena (52 mg LNG, 5 years), Kyleena (19.5 mg LNG, 5 years), Liletta (52 mg LNG, 6 years), Skyla (13.5 mg LNG, 3 years). * Duration of action: 3-6 years depending on type. #### Mechanisms of Action * **Cu-IUCDs:** * **Primary:** Local sterile inflammatory reaction in the endometrium, producing cytotoxic effects on sperm and ova. Copper ions enhance this reaction, impairing sperm viability and function (motility, capacitation). * **Secondary:** Prevents fertilization and implantation by making the uterine environment unsuitable. * **LNG-IUS:** * **Primary:** Localized release of levonorgestrel. * Thickens cervical mucus (inhibiting sperm transport). * Thins endometrial lining (preventing implantation). * Suppresses ovulation in some women (less consistently than OCPs, more common with higher dose Mirena than lower dose Skyla/Kyleena). ### IUCDs: Non-Contraceptive Benefits & Risks #### Non-Contraceptive Benefits * **LNG-IUS:** * Significantly reduces menstrual blood loss (treatment for menorrhagia). * Reduces dysmenorrhea. * Treatment for endometriosis and adenomyosis (reduces pain). * Endometrial protection during estrogen therapy (e.g., HRT). * Reduced risk of endometrial hyperplasia/cancer. * **Cu-IUCD:** * No systemic hormonal effects. * Can be used as emergency contraception (up to 5 days post-unprotected intercourse, most effective EC). #### Risks & Side Effects * **Common Side Effects:** * **Cu-IUCD:** Increased menstrual bleeding, longer periods, increased dysmenorrhea (often worse in first few cycles, can persist). * **LNG-IUS:** Irregular bleeding/spotting (especially in first 3-6 months, often leading to amenorrhea later), headache, breast tenderness, mood changes, acne (less common and milder than OCPs). * **Serious Risks (Rare):** * **Uterine Perforation:** Occurs in 1-2 per 1000 insertions. Risk factors: inexperienced clinician, postpartum/lactating women. Most perforations are fundal. * **Expulsion:** 2-10% in the first year, higher if inserted postpartum/post-abortion. * **Pelvic Inflammatory Disease (PID):** Risk is primarily in the first 20 days post-insertion (due to introduction of bacteria from cervix). No increased risk after 20 days compared to non-users. * **Ectopic Pregnancy:** If pregnancy occurs with an IUCD in situ, the risk of it being ectopic is higher (relative risk) than for a pregnancy without an IUCD. However, the absolute risk of pregnancy (and thus ectopic pregnancy) is very low due to high IUCD efficacy. * **Infection:** Localized infection at insertion site. * **Lost Strings:** Can indicate expulsion, perforation, or simply retraction into the cervical canal. Requires investigation (ultrasound, hysteroscopy). #### Contraindications (WHO MEC) * **Category 4 (Absolute Contraindication):** * Current pregnancy * Unexplained vaginal bleeding (prior to evaluation) * Current PID or puerperal sepsis * Current STI (gonorrhea, chlamydia) or other current reproductive tract infection * Known or suspected uterine or cervical cancer * Distorted uterine cavity (e.g., large fibroids, congenital anomalies) * Breast cancer (for LNG-IUS, current or past 5 years) * Severe thrombocytopenia or coagulopathy (for Cu-IUCD, due to bleeding risk) * **Category 3 (Risks generally outweigh benefits):** * HIV infection (well-controlled HIV is Category 2 for LNG-IUS, Category 3 for Cu-IUCD per some guidelines) * High individual risk of STI exposure * History of breast cancer (for LNG-IUS, if >5 years since remission) * Uterine fibroids not distorting the cavity (Category 2, can be used with caution) ### IUCDs: Insertion & Management #### Pre-Insertion Assessment * **History:** Menstrual history, parity, STI risk, medical conditions, medication. * **Physical Exam:** Pelvic exam, bimanual exam to assess uterine size, shape, position. * **STI Screening:** Recommended for women at risk of STIs. * **Pregnancy Test:** Essential prior to insertion. * **Counseling:** Discuss method efficacy, side effects, risks, warning signs, follow-up. #### Insertion Procedure * Performed by trained healthcare provider. * Usually done during menses or shortly after (cervix is softer, confirms non-pregnancy). Can be inserted at any time if pregnancy is excluded. * **Steps:** 1. Bimanual exam. 2. Speculum insertion, cervical cleansing. 3. Local anesthetic (optional, but recommended for nulliparous or anxious women). 4. Uterine sounding to measure depth and direction. 5. IUCD insertion using applicator. 6. Trimming of strings (approx. 2-3 cm from cervix). 7. Confirmation of placement (palpation of fundus, ultrasound if needed). #### Post-Insertion Care & Follow-up * **Immediate:** Advise on potential cramping, spotting. Provide analgesia. * **Follow-up:** Usually 4-6 weeks post-insertion to check strings and address concerns. * **String Check:** Teach patient how to feel for strings regularly (monthly after menses). * **Warning Signs:** Severe pain, fever, abnormal discharge, heavy bleeding, missing strings. #### Removal * Can be removed at any time by a healthcare provider. * Fertility returns rapidly after removal. * If patient desires continued contraception, new IUCD can be inserted at same visit. #### Emergency Contraception with Cu-IUCD * Highly effective when inserted up to 5 days after unprotected intercourse. * More effective than oral EC pills. * Offers ongoing contraception. ### OCPs vs. IUCDs: A Comparative Analysis | Feature | Combined Oral Contraceptives (COCs) | Levonorgestrel-IUS (LNG-IUS) | Copper-IUCD (Cu-IUCD) | | :--------------------- | :------------------------------------------------------------------------ | :------------------------------------------------------------------ | :------------------------------------------------------------- | | **Efficacy (Perfect Use)** | 99.7% | >99.8% | >99.4% | | **Efficacy (Typical Use)** | 91% | >99.8% | >99.4% | | **Duration of Action** | Daily | 3-6 years | 10-12 years | | **Mechanism** | Ovulation suppression, cervical mucus thickening, endometrial thinning | Cervical mucus thickening, endometrial thinning, partial ovulation suppression | Sterile inflammatory reaction, spermicidal, prevents implantation | | **Hormonal?** | Yes (Estrogen & Progestin) | Yes (Levonorgestrel) | No | | **Administration** | Oral, daily | Uterine insertion by provider | Uterine insertion by provider | | **Return to Fertility**| Rapid (within 1-3 months) | Rapid (within 1-3 months) | Rapid | | **Non-Contraceptive Benefits** | Reduced dysmenorrhea/menorrhagia, acne, PCOS, reduced cancer risk (ovarian, endometrial) | Reduced menorrhagia/dysmenorrhea, endometriosis, endometrial protection | Emergency contraception, no systemic hormones | | **Common Side Effects**| Nausea, breast tenderness, headache, breakthrough bleeding, mood changes | Irregular bleeding/spotting (initial), amenorrhea (later), headache, mood changes | Increased menstrual bleeding/cramping | | **Serious Risks** | VTE, MI, Stroke, HTN, Gallbladder | Perforation, expulsion, PID (initial), ectopic pregnancy (if pregnant) | Perforation, expulsion, PID (initial), ectopic pregnancy (if pregnant), heavier menses | | **Contraindications** | VTE, MI, Stroke, migraine with aura, uncontrolled HTN, current breast cancer, smoking >35yo | Current pregnancy, distorted uterus, current PID/STI, breast cancer | Current pregnancy, distorted uterus, current PID/STI, severe bleeding disorders | | **STI Protection** | No | No | No | | **Patient Adherence** | High daily adherence required | No daily adherence required | No daily adherence required | ### Special Considerations in Contraception #### Postpartum Contraception * **Breastfeeding:** * **COCs:** Generally avoided for first 6 weeks postpartum due to VTE risk and potential impact on milk supply. After 6 weeks, low-dose COCs can be considered if milk supply is well-established. * **POPs:** Can be started immediately postpartum and are preferred for breastfeeding women as they don't affect milk supply. * **LNG-IUS/Cu-IUCD:** Can be inserted immediately postpartum (within 10 minutes of placental delivery) or delayed to 4-6 weeks postpartum. Increased risk of expulsion with immediate insertion. * **Non-Breastfeeding:** * **COCs:** Can be started 3 weeks postpartum (due to VTE risk). * **POPs/LNG-IUS/Cu-IUCD:** Can be started immediately postpartum. #### Contraception for Adolescents * **LARCs (IUCDs, Implants):** Recommended as first-line due to high efficacy and no adherence issues. * **COCs:** Effective but require consistent adherence, which can be challenging for adolescents. * Counseling must be age-appropriate, address STI prevention, and ensure confidentiality. #### Contraception for Perimenopausal Women * Contraception is needed until 1 year after the last menstrual period (FMP) if >50 years old, or 2 years after FMP if 35 kg/m2 for some formulations). LARCs are excellent options. * **Diabetes:** COCs can be used if no vascular complications. LARCs are preferred. * **Hypertension:** COCs contraindicated if uncontrolled HTN (≥160/100). If controlled (140-159/90-99), Category 3 for COCs. POPs, LNG-IUS, Cu-IUCD are safe. * **Migraine:** COCs contraindicated if migraine with aura (any age) due to increased stroke risk. POPs, LNG-IUS, Cu-IUCD are safe. If migraine without aura and 35, Category 3. * **Solid Organ Transplant:** Complex, often requires specialist consultation. Immunosuppressants can interact with hormonal contraceptives. IUCDs are often preferred. ### Future Directions in Contraception Research continues to evolve, focusing on improving efficacy, safety, user convenience, and expanding options. #### Novel Hormonal Methods * **New Estrogens:** Estetrol (E4) in COCs, offering a potentially more favorable safety profile. * **New Progestins:** Development of progestins with specific receptor affinities for targeted effects. * **Non-Estrogen COCs:** Efforts to develop COCs that do not contain estrogen, potentially reducing VTE risk. #### Non-Hormonal Methods * **Pill for Men:** Research into reversible male contraception targeting sperm production or function. * **Vaginal pH Modulators:** Gels that lower vaginal pH to immobilize sperm. * **Immune-based Contraception:** Vaccines targeting sperm or egg proteins. #### Improved Delivery Systems * **Longer-acting Implants/IUCDs:** Extending the lifespan of existing LARCs. * **On-demand Vaginal Rings:** User-inserted rings for short-term use. * **Microbicides with Contraception:** Dual-purpose products for STI prevention and contraception. #### Personalized Contraception * **Genetic Profiling:** Tailoring contraceptive choice based on individual genetic risk factors for VTE or other adverse effects. * **Digital Health Tools:** Apps and wearables for improved adherence, symptom tracking, and personalized advice. ### Conclusion OCPs and IUCDs are highly effective and widely used contraceptive methods, each with distinct advantages, mechanisms, and side effect profiles. A thorough understanding of their pharmacology, benefits, risks, and contraindications, guided by tools like the WHO Medical Eligibility Criteria, is essential for every healthcare provider. Patient-centered counseling, considering individual medical history, lifestyle, and preferences, is paramount to ensure optimal contraceptive choice and adherence, thereby promoting reproductive health and well-being.